Evolution of Enzyme Requirements for Human Islet Isolation
Robert C. McCarthy * ![]() , Michael L. Green
, Michael L. Green ![]() , Francis E. Dwulet
, Francis E. Dwulet ![]()
VitaCyte LLC, Indianapolis, Indiana, United States of America
* Correspondence: Robert C. McCarthy ![]()
Academic Editor: Kåre I. Birkeland
Received: September 24, 2018 | Accepted: November 05, 2018 | Published: November 8, 2018
OBM Transplantation 2018, Volume 2, Issue 4 doi:10.21926/obm.transplant.1804024
Special Issue: Current Advancement of Islet Cell Transplantation in the Treatment of Diabetes Mellitus
Recommended citation: McCarthy RC, Green ML, Dwulet FE. Evolution of Enzyme Requirements for Human Islet Isolation. OBM Transplantation 2018; 2(4): 024; doi:10.21926/obm.transplant.1804024.
© 2018 by the authors. This is an open access article distributed under the conditions of the Creative Commons by Attribution License, which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is correctly cited.
Abstract
Islet transplantation is becoming an established treatment option for managing a subset of adult patients who have type 1 diabetes mellitus. The success of this procedure is dependent upon the recovery of a sufficient number of functional human islets from donor organs for subsequent transplant. Here, the use of optimized bacterial collagenase-neutral protease enzyme mixtures has been shown to affect the yield and quality (defined by viability and glucose-stimulated insulin secretion) of islets recovered from human pancreata. However, few reports provide a systematic approach to correlate the biochemical characteristics or dose of collagenases and proteases to the recovery of functional islets. The focus of this review is to close this gap. The review summarizes five key advances toward an understanding of how Clostridium histolyticum collagenase and bacterial neutral proteases support the release of islets from human pancreata and key collagenase biochemical characteristics that lead to higher human islet yields. This information provides a foundation to develop a model of enzymatic tissue dissociation that can serve as a guide in assessing the effectiveness of collagenase or neutral protease enzymes used for human islet isolation. One key conclusion is the importance of excess amounts of collagen degradation activity in the islet isolation procedure to ensure effective degradation of native collagen. If achieved, the selection and dose of neutral protease become the primary factors that determine the maximal release of islets from tissue. Maximal collagen degradation activity is achieved when an appropriate dose of C. histolyticum collagenase products containing primarily intact class I and class II collagenase are used in the isolation procedure. The review concludes by presenting future directions for research to develop new insights into enzymatic tissue dissociation with the goal of improving the robustness of the islet isolation process.
Keywords
Collagenase; neutral protease; tissue dissociation; human islet isolation; islet isolation; islet transplantation; cell isolation; Col H; Col G
1. Introduction
Human islet allotransplantation requires recovery of sufficient numbers of functional islets from human pancreata for subsequent transplantation. Purified collagenase-protease enzyme mixtures have been used to isolate islets for nearly all clinical human islet isolations since 1999 [1]. The success of the Edmonton Protocol for using islet transplantation to mitigate the risk of hypoglycemic unawareness in adult type I diabetic patients [2] led to funding the Clinical Islet Transplantation (CIT) consortium trial in 2004 [3]. This funding focused on standardizing the human islet isolation procedure to be compliant with GMP Guidelines. These to islets were used in clinical trials designed to assess the benefits of islet transplantation to manage adult type 1 diabetes patients. The ultimate goal for this funding was to enable institutions who participated in the trial to submit their data to the United States Food and Drug Administration (FDA) to gain regulatory approval for this treatment.
For many years, the sole product used for human and animal islet isolation and sold by numerous commercial suppliers was crude or enriched collagenase, a minimally processed Clostridium histolyticum culture supernatant. In the mid 1990’s, Liberase™ HI was developed at Boehringer Mannheim Biochemicals to overcome the lot to lot variability found in these collagenase products. The success of Liberase HI was dependent on the purity and performance of the collagenase and protease enzymes required to release islets from tissue. In 2007, the National Institutes of Health (NIH) forbade the use of Liberase HI in any NIH sponsored clinical trials because of the potential risk for transmission of spongiform encephalopathy [4]. The CIT switched to using the Nordmark/Serva purified collagenase and enriched neutral protease products, but some laboratories had difficulties to achieve islet isolation results similar to those obtained with Liberase HI [5,6,7]. In 2008, The CIT adopted VitaCyte’s and Roche’s purified collagenase and protease enzymes that had minimal risk for passing Transmissible Spongiform Encephalopathy (TSE) to patients who participated in the trial. Roche developed a new Liberase product (Liberase™ MTF) to replace Liberase HI, discontinued in 2008.
Collagenase-protease enzyme mixtures are critical reagents for islet isolation since their effectiveness to recover human islets determines if an islet transplantation procedure is performed. A recent paper summarizing the manufacture of human islets for use in the CIT-07 clinical trial showed only 52% of the islet isolations manufactured by eight centers provided a sufficient number of islets for subsequent transplantation [8]. The success rate from each center ranged from 24.3 to 89.5%. The failure to obtain a sufficient number of islets for transplantation may curtail the application of this therapy to treat diabetic patients. The cost of one islet isolation can be as high as 20,000 to 37,000, in the United Kingdom and the United States (U.S.), respectively [9,10]. These costs do not include that charged for the organ by the U.S. organ procurement organization or the associated physician's fees and hospital costs related to treatment. It’s imperative that the best knowledge be applied to human islet isolation to reduce the percentage of failed islet isolations, further reducing costs and for maintaining this treatment as a viable option for adult type 1 diabetic patients.
The key challenge for the manufacture of GMP human islets for clinical allotransplantation is to define critical parameters that affect recovery of sufficient numbers of functional islets from human pancreas for subsequent transplantation. Enzyme mixtures used for human islet isolation are a critical process parameter for successful islet isolation, yet few systematic studies have analyzed the influence of collagenase and protease activity on human islet yield and function. The poor characterization of the enzymes used in many studies often confounds interpretation of the results. The purpose of the current report is to review published literature that provides guidance on key characteristics of enzymes that are likely to improve human islet yield, viability, and function and is broken down into sections as follows.
-
Successful human islet isolation requires the use of defined collagenase-protease enzyme mixtures
-
Intact class I (C1) and class II (C2) collagenase should be used to ensure maximal islet recovery at the lowest dose of enzyme
-
Model of tissue dissociation: defining the roles of collagenase and protease in releasing cells from tissue
-
Validating the premises of the model by using the design of experiment approach
-
Different C1:C2 ratios within a defined range do not influence islet yields
-
Differences in the efficiency of purified natural and recombinant collagenase to recover functional human islets
-
Selection and dose of neutral protease is critical for successful human islet isolation
-
Perspectives and future directions for research
This review discusses selected aspects of the fundamental biochemical characteristics and assays used to assess Clostridium histolyticum collagenase and other bacterial neutral proteases used to isolate islets for transplantation. For additional information on the biochemical characteristics of these enzymes, please refer to an earlier review [11]. A recent review also discusses the use of enzymes for human islet isolation [12] but it does not focus on associating the biochemical characteristics of these enzymes to human islet yield or function.
2. Successful Human Islet Isolation Requires the Use of Defined Collagenase-Protease Enzyme Mixtures
The launch of Liberase™ HI Purified Enzyme Blend product at the end of 1994, was a breakthrough reagent that directly contributed to the success of islet transplantation [13,14]. Before the introduction of the Liberase HI product, most laboratories dedicated significant resources to screen different lots of crude or enriched collagenase for their effectiveness to isolate porcine or human islets. In the early 1990’s, Boehringer Mannheim Biochemicals’ Collagenase P became the favored product for this application, primarily driven by Camillo Ricordi, who used this product to isolate islets for subsequent transplantation [15]. However, Collagenase P had several limitations. Only about one-third of the lots were effective for human islet isolation and these “good lots” were stable for at most 18 months when they became ineffective in recovering human islets [16]. Moreover, the enzyme composition of good lots was unknown, making it difficult to define the unique characteristics of an exceptional lot of product.
Camillo Ricordi was the individual primarily responsible for initiating the development of Liberase HI at Boehringer Mannheim Biochemicals. His use of Collagenase P to prepare cells for clinical islet isolation led the company to send representatives to Pittsburgh to meet with him to discuss the clinical application of collagenase for islet transplantation. Ricordi later visited Indianapolis and met with senior R&D staff at Boehringer Mannheim. This meeting led to funding a project to develop a purified enzyme mixture for use in human islet isolation. The project started at the end of 1992 and the Liberase HI product released for sale in North America in November 1994.
Liberase HI was the first purified enzyme formulation developed for a specific cell isolation application. This composition reflected the conclusions from earlier studies that indicated collagenase and protease enzyme activities were the key enzymes required to release cells from tissue [17,18,19]. The initial report described the effectiveness of Liberase HI to isolate islets from 26 research donor pancreata [20]. Subsequent reports showed the superiority of this reagent over crude or enriched collagenase [21,22] but average islet yields were 56% (n=50) and 61% (n=13) of those obtained in the initial report [20]. Several factors may be responsible for this difference. The first study [20] did not measure islets purified after density gradient purification but quantitated their kinetic release from the tissue using a custom imaging system that detected islets stained with N-(6-methoxy-8-quinolyl)-p-toluenesulfonamide (TSQ) [23]; and the mean cold ischemia time of the pancreata used in the initial report was shorter (4.9 hours) than the latter two reports (8.8 hours).
Liberase HI was the primary purified enzyme mixture used for human islet isolation from 1994 to 2007. Liberase HI served as a benchmark for comparison of all other purified enzyme products used to isolate human islets [7,24,25,26]. For many years, nearly all laboratories isolating islets for clinical transplantation used this product, but the specifics of its development and changes made to the enzyme formulation over its product lifetime were unknown to many users. These include the rationale for selecting the collagenase composition and protease used in the product; and changes to the enzyme formulation from 1994 to 2007. The review of Liberase HI summarized below shows that at the time of product development, there was rudimentary knowledge of the key enzymes responsible for tissue dissociation and no understanding of how these enzymes contributed to successful islet recovery. Development of Liberase HI represented the first commercial use of defined enzyme mixtures for cell isolation and provided the foundation to build additional knowledge which is the subject of this review.
2.1 Development of Liberase HI
The key factor responsible for the successful development of the Liberase HI product was feedback from customers who evaluated selected lots of Collagenase P for recovery of porcine or human islets. Customers categorized each lot as “good” or “bad” for this application. Table 1 summarizes the ranges of enzyme activities found in “good lots” of Collagenase P. Earlier reports showed that successful cell isolations required the use of collagenase and protease activity [17,18,19]. These observations led the Liberase project team to develop assays to measure collagenase and neutral protease activity using Pz peptide and fluorescein isothiocyanate-casein substrates, respectively [27,28].
Table 1 Enzyme activities per 1 g bottle from “good lots” of Collagenase P 1990-1993

At the time of product development two isoforms of C. histolyticum collagenase, C1 and C2 collagenase, were defined by differences in substrate specificity. Purified C1 collagenase has low specific peptidase activity (i.e., Wunsch U/mg protein), and high specific gelatinase activity (U/mg protein) whereas the converse is true for C2 collagenase [30]. Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) of different purified fractions of crude collagenase identified six different forms of the enzyme that ranged from 80—130 kDa [31].
Analytical anion exchange chromatography procedures were adopted to enable quantitative assessment of the relative concentrations of C1 and C2 [32]. Figure 1 compares anion exchange chromatography profiles of Collagenase P used as the raw material for collagenase purification and Liberase HI. The appearance of multiple C1 peaks, (C1, C1b, C1c, C1d, C2b), indicated degradation of C1 but the significance of this observation was unknown at the time.
The initial strategy to manufacture Liberase HI was to isolate C. histolyticum collagenase and neutral protease enzymes from Collagenase P. As shown in Figure 1, Collagenase P contained predominantly single C1 and C2 peaks whereas Liberase HI gave single symmetrical C1 and C2 peaks after analysis by analytical anion exchange chromatography. The high clostripain activity found in most lots of Collagenase P was problematic as this enzyme is a common contaminant found in purified collagenase and difficult to remove [33]. Variable contamination of collagenase with clostripain led the initial formulation of Liberase HI to contain a fixed amount of clostripain (7,000 BAEE U/bottle) to ensure consistency of the neutral protease activity [34].
Purification of C. histolyticum neutral protease (CHNP) was difficult because its activity was unstable upon storage. Purified thermolysin or Dispase™ was used in place of CHNP, based on the assumption that any other neutral proteases would be as effective as CHNP for human islet isolation. Purified thermolysin and Dispase were found to have acceptable purity and stability. Thermolysin was used instead of Dispase because of cost considerations. Dose-response experiments correlating Liberase HI mass to islet yields showed the use of 0.75, 1.5, or 3.0 mg of Liberase HI per mL of solution did not affect the release of human islets from tissue [20]. The final enzyme composition of version 1 of Liberase HI product was about 1600 Wunsch U of purified collagenase and about 90,000 FITC-casein units per bottle. Seven mg of purified thermolysin and 7,000 BAEE U of clostripain contributed to this FITC casein activity.
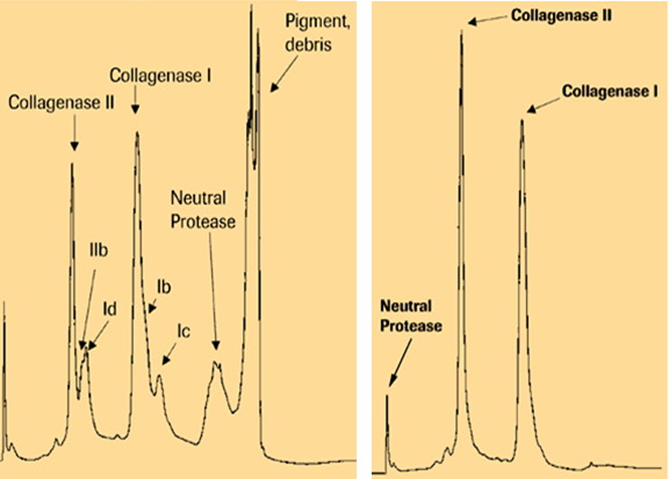
Figure 1 Analytical anion exchange chromatographic analysis of Collagenase P (A) and Liberase HI (B). Additional forms of collagenase are designated by class and alphabetical lower case. ©2000 All rights reserved, Roche Diagnostics Corporation, USA, reprinted with permission.
Table 2 summarizes further changes to the Liberase HI formulation from 1994 to 2008. Version 1 was considered a first generation product since the project team realized that further improvements would be made at a later date to improve the consistency of the enzymes and to adapt the enzyme composition to meet user’s needs. An additional column chromatography step was added to the collagenase purification procedure to minimize clostripain contamination in version 2 Liberase HI. Clostripain is a proteolytic enzyme with trypsin-like activity and requires a reducing environment for maximal activity [29]. Under normal conditions, the majority of the sulfhydryl groups are oxidized, with only about 10% of the clostripain active. The enzyme activity is measured by the hydrolysis of N-alpha-benzoyl-L-arginine ethyl ester (BAEE), a peptide substrate used to detect trypsin activity. Removal of clostripain improved product consistency. Additional thermolysin protease activity was added to the Liberase HI product to maintain the same amount of neutral protease activity found in version 1 of the product.
Feedback from participants who were planning to participate in the Immune Tolerance Network (ITN) clinical trial led to changes in enzyme composition in version 3 Liberase HI. The goal of the ITN trial was to have other islet transplant centers verify the results from the Edmonton Protocol [35]. This collaborative effort between scientists at Roche Applied Sciences (formerly, Boehringer Mannheim Biochemicals) and the ITN participants led to a decrease in the neutral protease activity used in the Liberase HI formulation.
Table 2 Multiple versions of Liberase HI

The changes in version 4 of Liberase HI were due to unintended consequences of a change in the chromatography resin used to capture collagenase in the first chromatographic step of the purification procedure. Lakey summarized the steps used to purify collagenase for Liberase HI at an Islet Resource Center Workshop in 2005 [36]. After this change, more degraded C1 was present than found in the three earlier versions of Liberase HI. This issue was unresolved, and the problem persisted from about 2003 to the discontinuation of the manufacture of Liberase HI in 2008. Several reports identified this change with newer lots of the Liberase HI product [37,38] with one indicating that it may be due to a change in the manufacturing procedure [37]. At the time of this change, none of the key members of the original Liberase project team who had responsibility for developing the enzyme purification process were employed by Roche Applied Sciences.
In retrospect, several critical factors fell into place during the development of Liberase HI at Boehringer Mannheim Biochemicals. Collagenase P was an excellent raw material that led to the development of cost-effective enzyme purification process. The proposed enzyme formulations were rapidly tested by having a porcine islet isolation group as part of the Liberase project team. This team rapidly assessed the performance of these enzyme mixtures by assessing their effectiveness to recover porcine islets from market weight pigs sourced from a small pig farm in central Indiana. Unbeknownst to the team, recovery of high numbers of intact islets from market weight pigs was a unique observation. About a year after the launch of version 2 of Liberase HI, porcine islet isolations were re-started, and the project team found that earlier porcine islet yields were difficult to replicate. Further experiments showed that high yields of porcine islets correlated with the pedigree of the boars who sired the pigs that were the source of pancreata for islet isolation [39].
Jonathan Lakey’s contribution to the development of the Liberase products cannot be underestimated. Jonathan was a postdoctoral fellow in Indianapolis at the time the project team validated the Liberase HI formulation for human islet isolation. His efforts to procure human research organs and the transitioning of the porcine islet isolation team to process human pancreata were critical to the success of the project.
The challenges faced by the project team that developed the Liberase product line should not be understated. C. histolyticum culture supernatants contain proteases that led to degradation of collagenase, a problem that contributes to the lot to lot variability typically found in crude and enriched collagenase products. Schemes had to be developed to limit this degradation. Next, there was little understanding of the basic biochemistry of collagenase. Van Wart and colleagues identified six different forms of C. histolyticum collagenase, but it was uncertain whether each form reflected proteolysis, differential translation, or expression of separate genes [40]. Moreover, there was little understanding of the mechanism of how collagenase and protease released cells from the extracellular matrix. Despite this limited knowledge, the launch of Liberase HI laid the groundwork to make further improvements to collagenase-protease enzyme mixtures. These improvements are the subject of the remainder of this review.
3. Intact Class I (C1) and Class II (C2) Collagenase Should be Used to Ensure Maximal Islet Recovery at the Lowest Dose of Enzyme
The unintentional consequences of a resin change used in the collagenase purification process led to significant changes in the ability of Liberase HI to recover human islets. Lakey and Barnett et al. showed the appearance of multiple C1 peaks in Liberase HI correlated with their date of manufacture [36,37]. They believed this was due to a change in the manufacturing process and not in the instability of the product during storage. They developed a kinetic, fluorescent microplate assay using a commercial substrate, fluorescein-labeled DQ gelatin conjugate, to show that lower gelatinase activities found in sub-optimal lots of Liberase HI contained multiple C1 peaks as detected by anion exchange chromatography. Further analysis of four different lots of Liberase HI used to recover human islets from 12 isolation procedures were categorized as “successful” (> 300,000 IEQ/pancreas) or “unsuccessful” (< 300,000 IEQ/pancreas). Comparison of the effectiveness of the enzymes used to recover these islets showed that lots with a higher specific gelatinase activity resulted in significantly more successful isolations when compared to those isolations that used low specific gelatinase activity.
A subsequent report confirmed Barnett’s et al. conclusions, showing variability in human islet yields when 169 islet isolations were performed using different lots of Liberase HI [38]. This retrospective analysis showed a significant decrease in pre and post-purification islet yields when two different lots of product were analyzed over a 15 month period, indicating loss of activity during storage.
Barnett’s et al. data indicated the presence of multiple C1 peaks appeared to correlate with decreased islet yields, but there was no mechanistic explanation of how the change in collagenase activity led to decreased islet yields [37]. The development of fluorescent microplate collagen degradation activity (CDA) assay that detected collagenase enzymes that could cut tropocollagen [41], the triple alpha-helical protein chain, a common motif found in all types of collagen [42], explained Barnett’s results. Analyses of peak fractions (C2, C1, C1b, and C1c) recovered after passing purified collagenase over an anion exchange chromatography column by SDS-PAGE or assay of collagen degradation activity (CDA), showed that the specific CDA correlated with the molecular form of collagenase as defined by Matsushita et al. [43]. Figure 2 shows the three-dimensional protein domain structure of C1 and C2 collagenase. C1 or C2 has four protein domains: an amino-terminal catalytic domain, one or two linking domains (function unknown) that have homology to polycystic kidney disease-like domains [44], and one or two collagen binding domains. Functionally active collagenase, defined by the ability to degrade tropocollagen, must contain a catalytic domain and at least one collagen binding domain. The three functionally active forms shown on the left side of Figure 2 are:
-
intact C1 (C1116 kDa) containing a catalytic domain, a linking domain, and two collagen binding domains;
-
intact C2 (C2114 kDa) containing a catalytic domain, two linking domains, and one collagen binding domain;
-
and truncated C1 (C1100 kDa) containing three domains, a catalytic domain, a linking domain, and only has one collagen binding domain.
Proteolysis at a 24 amino acid random coil segment that connects the two collagen binding domains on intact C1 led to the formation of a truncated C1 form. These shorter molecular forms contain one collagen binding domain. This proteolysis likely occurs during the fermentation and/or the enzyme purification process. These analyses showed that these truncated forms of C1 (C1b, and C1c), had lower specific CDA. Later studies showed that the C1c form had 12 more amino acids than the C1b form when these purified forms were sequenced using mass spectrophotometry [45]. This observation is the likely reason for the different elution patterns off the anion exchange chromatography column.
C1 or C2 without a collagen binding domain are termed non-functional collagenase because they cannot degrade tropocollagen (right side of Figure 2). These two forms are common non-functional, degraded forms of collagenase but any form of C1 < 85 kDa or C2 < 100 kDa created by further proteolysis of these enzymes cannot digest tropocollagen. It is important to note when SDS-PAGE is used to characterize collagenase, it is subject to artifacts. Contaminating clostripain or other proteases can degrade collagenase rapidly during the sample preparation process. The addition of 10 mM EDTA into the sample before heat denaturation of the protein eliminates this artifact [31].
The table at the bottom of Figure 2 shows the correlation of enzyme activities to the molecular form of C1 or C2 collagenase. All forms of collagenase with a functional catalytic domain can digest gelatin or cleave collagen-specific peptides (Pz or FALGPA peptide). These assays detect both functional and non-functional forms of collagenase. If the catalytic domain is damaged or lost, then no gelatinase or peptidase enzymatic activity is detected. Only those forms of collagenase with a functional catalytic domain and at least one collagen binding domain have CDA. Intact C1 (C1116 kDa) with two collagen binding domains had about a 7 to 9 fold higher specific CDA than the molecular forms containing a single collagen binding domain: truncated C1 (C1100 kDa) or intact C2 (C2114 kDa) [41]. These data indicate that enzyme preparations with more intact C1 may be more efficient in human islet isolation than preparations containing truncated C1.
Balamurugan et al. [7] confirmed the validity of the assumption that intact C1 is more effective in human islet recovery than truncated C1. They correlated biochemical analysis of collagenase to its effectiveness to recover high numbers of human islets. Three different lots of purified collagenase from Roche Applied Sciences (Liberase HI), Nordmark/Serva (NB-1 Collagenase), and VitaCyte (CIzyme Collagenase HA) were analyzed by anion exchange chromatography and for specific CDA (CDA U/mg) using the assay cited above [41]. These results supported the observations made during the development of the CDA assay: the higher amount of the C1116 kDa as a percentage of total collagenase, the higher the CDA specific activity of the collagenase preparation.

Figure 2 Functional and non-functional forms of C. histolyticum collagenase and enzyme activity profile. The 3-dimensional structures are individual domains of class I C. histolyticum collagenase obtained from the Protein Database [46,47,48]. For illustrative purposes, the class I domains are also used to represent class II collagenase domains. The collagen degradation activity assay used to assign relative activity [41].
Further experiments compared human islet yields from isolations using either Nordmark/Serva NB-1 Collagenase-NB Protease enzyme mixtures (n=27: 17 research, 10 clinical) or VitaCyte Collagenase HA-Thermolysin (n=14: 4 research, 10 clinical) enzyme mixtures. The results showed that the Nordmark/Serva enzymes gave significantly longer digest times and significantly lower post-digest IEQ/pancreas, post-digest IEQ/g pancreas, post-purification IEQ/pancreas, and post-purification IEQ/g pancreas when compared to isolations using VitaCyte’s enzymes. The lower islet yields obtained when using the Nordmark/Serva products reflected the lower percentage of these islet preparations used for subsequent transplant. Twenty (2/10) and forty percent (4/10) of the islet preparations isolated using the Nordmark/Serva and the VitaCyte’s enzyme, respectively, were used for clinical islet transplantation [7].
Results reported by the Clinical Islet Transplant Registry, an international registry that collects and analyzes data from centers that perform allo-islet transplantation, showed that the lowest number of islet transplants performed during the period from 2001 to 2013 was in 2007. The likely reason for this low number was the forced change in 2007 from use Liberase HI to the Serva enzymes at centers participating in the CIT study. This change occurred because of the potential risk of TSE transmission when using Liberase HI for islet isolation [4].
The number of islet isolations reported to the registry increased from 2008 to 2013, reflecting funding for islet transplantation from the CIT. An increased percentage of these isolations used the Nordmark/Serva enzymes where many laboratories made adjustments to their islet isolation procedure based on the different characteristics of these enzymes when compared to isolations using Liberase HI.
Although the NB-1 Collagenase and NB Protease continues to be used by many laboratories, one group found that those lots with higher amounts of clostripain activity gave higher human islet yields [49]. Another group found the addition of clostripain to Collagenase HA/thermolysin enzyme mixtures increased islet yields [50]. The relevance of adding clostripain to purified collagenase-protease enzyme mixtures will be discussed in more detail below.
A direct comparison of the effectiveness of intact or truncated C1 to recover islets from porcine pancreas was reported recently [51]. Two recombinant C. histolyticum collagenase-BP Protease enzyme mixtures were prepared that differed only in the molecular form of C1 (intact or truncated C1- rC1) and assessed for their ability to recover islets from an adult split porcine pancreas. All isolation used equivalent enzymatic activities: 25,000 CDA U/g trimmed tissue, 7.5 Wunsch U/g, and 12,000 FITC-BSA U/g for rC1116kDa or C1100kDa, rC2114kDa, and BP Protease, respectively. Isolations with rC1116kDa (n=8) and rC1100kDa (n=8) were equally stratified across splenic and duodenal/connecting lobes to avoid confounding factors. Analyses of results from these isolations showed that the percentage of undigested tissue, packed tissue volume per g pancreas and post-purification islet yield expressed as islet equivalents per g tissue were similar for isolations performed with either intact or truncated C1. Switch times were also similar. The striking observation was the total mass of intact C1 protein required to maintain a 25,000 CDA U/g trimmed tissue target averaged 37.6 ± 2.7 mg when compared to 504.6 ± 52.5 mg for truncated C1. This ≈ 13 fold difference in mass required to achieve this targeted CDA U/g tissue reflects differences in CDA specific activity: 5,050 for truncated C1 as compared to 87,500 CDA U/mg for intact C1.
The importance of intact C1 for successful human islet isolation raises questions regarding enzyme characteristics required for successful human islet isolation; a topic reviewed earlier [11]. Foremost among them is the reliance of Wunsch activity to determine the dose of collagenase used in human islet isolation. Wunsch activity is a reliable and convenient measure of collagenase activity, but its limitations are clear with the new knowledge of the structure-function of collagenase. The Pz peptide used in the Wunsch assay detects C2 collagenase activity. The difference in the specific Wunsch activity for C2 is at least 50 fold higher than the specific activity for C1. Moreover, Wunsch activity cannot distinguish between functional and non-functional forms of collagenase if functionality is defined by the ability of collagenase to degrade tropocollagen. Therefore, this reproducible and reliable assay has limited application as a surrogate measure of collagenase activity to predict the successful islet recovery as was shown in an earlier report from the University of Alberta [52].
There is a need to develop a transferable, verified, and validated CDA assay that would enable laboratories to qualify new lots of purified collagenase before use. The fluorescent, kinetic, microplate assay used in earlier experiments [41] is not amenable to transfer because fluorescent assays are difficult to replicate between laboratories. This difference is primarily due to the inability to calibrate the fluorescent measurements. Many different adjustments can be made to fluorescent instruments that are difficult to control between instruments. The answer to this limitation is to develop a spectrophotometric CDA assay that can be used to correlate CDA with islet yield. VitaCyte has developed a modified Mandl CDA assay that may prove to be a reliable surrogate collagenase assay for this application (F.E. Dwulet unpublished data). Further work must be performed to correlate the results from this assay to those obtained from the fluorescent, kinetic, microplate CDA.
Finally, what does the present biochemical analysis reveal about the characteristics of C2 collagenase? Unfortunately, there are no reports that use physicochemical analysis of C2 collagenase to assess enzyme integrity, so one has to assume that if the C1 is intact, then the C2 is also likely intact. SDS-PAGE may be used, but it is an imprecise method, subject to artifacts as noted above. Nor is a decrease in the specific Wunsch activity an indicator of degraded C2 simply because this activity is resistant to proteolysis [41]. A decrease in specific Wunsch activity more likely represents contamination of the C2 collagenase with degraded C1.
4. Model of Tissue Dissociation: Defining the Roles of Collagenase and Protease in Releasing Cells from Tissue
The information above shows the importance of using intact C1 collagenase in enzyme mixtures for porcine or human islet isolation, but no model explains the advantage of using this form of collagenase. A hypothetical model needs to provide practical direction as to how all three enzymes, C1 and C2 collagenase, and neutral protease interact to release cells from tissue. A simple model of how these enzymes may degrade the extracellular matrix is re-stated [11] and refined below, including additional data that support the primary premises of the model.
The key assumptions of the hypothetical model of enzyme-mediated tissue dissociation using C. histolyticum collagenase and neutral protease enzymes to release cells from the extracellular matrix (ECM) are summarized below.
1. The ECM is a complex mixture of macromolecules secreted by cells into intercellular spaces [53]
2. The ECM molecules include collagen, elastin, proteoglycans, glycosaminoglycans, and other proteins that anchor cells to the ECM [53]
3. Collagen fibrils (10-300 nm diameter) and fibers (0.5 to 3 μm diameter) are the predominant proteins in the matrix and are composed of collagen monomers (1.5 nm diameter) that associate in an overlapping pattern to form fibrils and the fibrils associate to form fibers [42]
4. Collagen fibrils and fibers are the skeletal backbones of the ECM, holding other macromolecules in a tight structure, resistant to proteolysis
5. Collagenase has a restricted substrate specificity: it degrades tropocollagen in many types of collagen (collagen types I, II, III, etc…) and degrades denatured collagen (i.e., gelatin)
6. Functional collagenase must bind to tropocollagen using its collagen binding domain before the catalytic domain can degrade this protease resistant, alpha triple helical protein structure
7. Functional and non–functional collagenase degrade gelatin
8. Some bacterial neutral proteases may degrade specific collagen types, but most proteases have broad specificity for degrading any protein, including gelatin
9. C1 and C2 collagenase, and neutral protease work synergistically to degrade extracellular matrix proteins, leading to the release of cells from tissue
Functional C. histolyticum collagenase initiates the tissue dissociation procedure by binding of the collagen binding domain to a site on tropocollagen. The model below emphasizes the role of C1 collagenase since more is known about this isoform than C2. However, it is clear from functional analysis of different mixtures of C1 and C2 using CDA assays and the use of BP Protease and a 30:70 recombinant collagenase C1:C2 enzyme mixtures that C2 collagenase plays an important role in collagen degradation and release of human islets from tissue [45,54].
Recombinant C1 collagenase has been shown to bind to tropocollagen within a fibril [55]. Once bound, collagenase’s catalytic domain can cut the collagen, leading to the unwinding of the triple helix to form denatured collagen (i.e., gelatin) that is degraded by functional or non-functional C1 or C2 collagenase or by neutral proteases. As the C1 collagenase continues to progress down the length of tropocollagen, the entire triple helical structure is degraded into smaller peptides [55]. The tight packing of tropocollagen in collagen fibrils appears to lead to binding of additional C1 collagenase to other tropocollagen molecules within the fiber, leading to rapid degradation of the collagen fibrils. It is thought that functional C2 collagenase binds in the middle portion of the tropocollagen molecule and works synergistically with C1, but a detailed mechanism of this role is unknown.
Collagen degradation leads to relaxation within the extracellular matrix, exposing protease sensitive sites in non-collagen, matrix proteins. These proteins include those that hold cells to the matrix (i.e., cell anchoring proteins) that include integrins, discoidin domain receptors, and syndecans. Once a sufficient number of these proteins are degraded, there is a maximal release of cells from the tissue. For human islet isolation, this is referred to as the “switch time” when the closed digestion circuit is opened and flushed with excess media with the cells collected into vessels and washed by centrifugation.
If this model is accurate, then excess collagenase activity is unlikely to affect the yield or function of cells recovered from tissue. Nearly all collagen types are found only in the extracellular matrix except types XIII, XVII, XXIII, and XXV that are membrane collagens that have restricted cell distribution [56]. Collagenase has been shown to diffuse into the interstices of exocrine and endocrine (i.e., islet) pancreas after retrograde infusion of the enzyme into the main pancreatic duct [57]. Collagenase could potentially digest the islets into cell clusters, but the digest is usually stopped before the fragmentation of the islet structure.
The exogenous proteases typically used are from Bacillus thermoproteolyticus (thermolysin), Paenbacillus polymyxa neutral protease (Dispase™, BP Protease), or C. histolyticum neutral protease. Prolonged cold ischemia storage time of pancreata (> 10 hours) or by contaminants in the collagenase or protease enzymes used for cell isolation leads to an increase in endogenous neutral protease enzyme activity [58,59,60]. This increase in activity is likely due to activation of serine protease enzymes from pancreatic proenzymes. One report showed the addition of purified collagenase alone was sufficient for islet release [60]. In these cases, there was prolonged cold ischemia time, and the purified collagenase contained contaminating clostripain. It is likely that the clostripain contaminant (trypsin-like protease) in purified collagenase contributed to the increase in endogenous protease activity by activating pancreatic proenzymes during the cell isolation process.
Several implications from the model above are:
• Intact forms of collagenase should be used for islet isolation to ensure efficient degradation of collagenase at the lowest dose of enzyme
• If intact collagenase is used in excess, the optimization of an enzyme formulation used for islet isolation should focus on the choice and dose of neutral protease.
5. Validating the Premises of the Model by Using the Design of Experiment (DOE) Approach
The hypothetical model of tissue dissociation presented above provides a framework to understand the contributions of C1, C2, and protease for successful human islet isolation. To validate this framework, rigorously characterized enzymes with minimal contaminants should be used in prospective human islet isolations, designed to test different premises of the model. Moreover, adopting a design of experiment approach provides the greatest insight into factors that affect islet isolation.
Table 3 summarizes the key differences in knowledge output between one factor at a time (OFAT) versus the design of experiment (DOE) approach. For both approaches, critical variables should be controlled before performing the experiment so that the results represent the effect of the independent experimental variable(s). In the OFAT approach, only one variable is changed at a time whereas for DOE, multiple variables can be assessed simultaneously using a design tied to statistical analysis. Greater insight into key factors that affect human islet isolation is likely to be obtained from using a DOE approach than one designed using OFAT. In a DOE, “all the observations can be used to estimate the effect and interaction (property of hidden replication), while typically only two of the observations in an OFAT experiment are used to estimate the effect of each factor” [61].
SAS (Cary NC) has developed a user-friendly software package (jmp™) that enables the power of DOE to be adopted by any laboratory that wants to improve the insights gained from performing expensive human islet isolation procedures.
The DOE approach was used by the Liberase project team to determine the optimal dose of collagenase and protease enzymes on porcine islet isolation [62]. Porcine islets were used in place of human islets for two reasons: the Liberase project team established a consistent model of islet recovery from market weight pigs, and at that time, porcine islets were commonly used to pre-screen new lots of collagenase before use for human islet isolation.
The DOE used for the Liberase project was a two factor, three-level test, with the islet isolations performed in duplicate. The design focused on determining an optimal composition and dose of enzymes for porcine islet isolation. The factors used in this experiment were purified C. histolyticum collagenase at a 60:40 C1:C2 ratio and the mass of purified Dispase. The dose was determined by pairing one of three different doses of collagenase activity (Wunsch U) with one of three different masses of Dispase used to isolate islets from intact porcine pancreas. An automated, fluorescent imaging system was used to remove any observer bias in performing the islet count [63]. Figure 3 shows a response surface plot that summarizes the experimental results. This plot showed the optimal dose for maximal islet release to be about 2200 Wunsch U per pancreas and 30 mg of BP Protease. This dose was confirmed by performing further isolations to validate this dose, and enzyme formulation. This enzyme composition was sold as Liberase PI from 1997-2008.
Table 3 Comparison of outcomes obtained from One Factor at a Time (OFAT) vs Design of Experiment (DOE) experimental designs [59]


Figure 3 Response surface plot of two factor three level design of experiment to determine optimal enzyme composition for isolation of porcine islets from market weight pigs. ©2000 All rights reserved, Roche Diagnostics Corporation, USA, reprinted with permission.
Figure 3 also shows the interaction between collagenase activity and protease mass. There is a small flat plateau, centered around 2200 Wunsch U and 30 mg of Dispase. The response surface is consistent with the hypothetical model of tissue dissociation presented above. From about 2000 to 2400 Wunsch U, the islet yield plateaus when using an optimal dose of protease in the isolation procedure. Once collagenase is below 2000 Wunsch U, islet yield decreases, indicating the importance of using excess collagenase in the islet isolation procedure. By contrast, the mass of neutral protease has a narrower plateau where the optimal dose is between 30-34 mg of Dispase. At a suboptimal dose, islet yield drops because there is insufficient protease to accelerate degradation of the extracellular matrix. At a supra-optimal dose, islets are likely fragmented, leading to lower islet yields.
An attempt was made to use DOE to determine an optimal dose for human islet isolation but was unsuccessful because of the variability between donors. The Liberase project team determined the formulation for version 1 Liberase HI from their analysis of good lots of Collagenase P [62].
6. Different C1:C2 Ratios within a Defined Range do not Influence Islet Yields
Review of the DOE approach by the Liberase project team provided partial validation of the major premise of the model presented above that neutral protease activity is likely a more critical factor to control than collagenase. Several factors limit the interpretation of the results from Liberase DOE. First, the enzyme dose was delivered to the entire organ and not adjusted by the mass of tissue. By basing the enzyme dose on the organ and not on the weight of trimmed pancreatic tissue, there is less control of a key variable since collagenase or protease activity per g pancreas can vary significantly between different isolations. Moreover, Wunsch activity does not measure functional collagenase activity since it reflects the activity of both functional and non-functional collagenase and is biased to detect only C2 collagenase.
A DOE designed experiment that directly answered the question of the influence of C. histolyticum C1 or C2 on human islet yield and function was reported recently [64]. Recombinant intact collagenase C1 (rC1116kDa) or intact C2 (rC2114kDa) used in these experiments were expressed separately as a cytoplasmic protein in E. coli from the gene sequence reported by Matsushita et al. [65]. The purified rC1116kDa and rC2114kDa used in a two factor, two-level DOE, with each condition performed in triplicate. The scientists who performed these isolations were blinded and unaware of the collagenase composition. For these experiments, islets were recovered from the body-tail portions of the pancreas. The head portion was used as a control to assess the effectiveness of the New Enzyme Mixture (NEM) to recover islets (details of this enzyme mixture described below). The doses of recombinant collagenase used were 100,000 or 200,000 CDA U of rC1, and 12 or 20 Wunsch U/g tissue of rC2, mixed with a fixed amount of purified BP Protease (23,400 NP U/g tissue). A wide range of C1:C2 ratios were used in these isolations: 55:45, 43:57, 38:62, and 27:73. The results from these experiments showed that the dose or ratio of C1 or C2 did not significantly influence islet yield (Figure 4) or islet function [64].
This experiment validates a major premise of the model presented above that human islet yield or function will be unaffected by a wide range of C1 or C2 collagenase activity. The key factor to control will be the selection and dose of the neutral protease, assuming that functional collagenase is in excess.

| rC2 + = 20 Wunch U/g tissue | rC2 - = 12 Wunsch U/g tissue |
| rC1 + = 200,000 CDA U/g | rC1 - = 100,000 CDA U/g |
Figure 4 Effect of C1 and C2 dose on recovery of human islets from the body-tail of the pancreas.
7. Differences in the Efficiency of Purified Natural and Recombinant Collagenase to Recover Functional Human Islets
The DOE studies above showed a wide range of rC1 and rC2 doses were successfully used to recover human islets [64]. The surprising finding from this experiment was the collagenase doses used in the DOE were 46 to 83 % (2.09-3.75 mg/g tissue) of the amount typically used for human islet isolation using natural, purified collagenase at a 60:40 C1:C2 ratio (4.5 mg/g tissue for NEM). Further studies by Balamurugan’s group at the University of Louisville showed that the lowest dose collagenase used in the DOE above (2.09 mg/g tissue at a 30:70 C1:C2 ratio) gave similar results when they were used to isolate human islets from intact pancreas (Figure 5) [54]. Further studies compared the effectiveness of purified natural or purified recombinant collagenase to isolate functional islets at a low (12 Wunsch U/g tissue) or a standard dose (20 Wunsch U/g tissue) of collagenase. BP Protease at 23,400 NP U per g tissue was used in all islet isolations. These results showed similar yield and function from islets isolated with 12 or 20 Wunsch U/ g tissue of recombinant collagenase or 20 Wunsch U/g of natural, purified collagenase. In contrast, there was a significantly lower islet yield when 12 Wunsch U/g tissue of natural, purified collagenase was used in the isolation procedure [54].
Functional analysis of the four different islet preparations by basal insulin release after 48 hours of culture showed a significantly lower insulin release when islets were isolated with the standard dose of natural collagenase (20 Wunsch U/g) when compared to islets isolated with the low dose of recombinant collagenase (12 Wunsch U/g). There were no significant differences in glucose-stimulated insulin release between the four groups [54].

Figure 5 Comparison of effectivenss of recombinant collagenase-BP Protease (BPP) vs purified natural collagenase-CHNP enzyme mixtures for human islet recovery. NEM: 20 Wunsch U Collagenase HA/g and 1.75 DMC U NB Protease/g pancreas; rC1rC2 + BPP (body/tail): four different rC1rC2 ratios + 23,400 NP U BPP/g pancreas; rC1rC2 + BPP (intact organ): 12 Wunsch U/g and 100,000 CDA U/g + 23,400 NP U BPP/g pancreas.
Presently, there is no explanation for the increased efficiency of recombinant collagenase to recover human islets or for the higher basal secretion of insulin after 48 hours of culture. There is a 4 and 13 amino acid sequence difference for C1 and C2 respectively when the sequences of the natural and recombinant collagenase were compared [45]. These differences represent a 0.4 and 1.3% change as a percentage of the total amino acid residues for C1 and C2, respectively.
8. Selection and Dose of Neutral Protease is Critical for Successful Human Islet Isolation
The information presented above emphasizes the importance of native collagen in the formation of a protease-resistant extracellular matrix. The model above proposes that C1 and C2 collagenase play a critical, but a very specific role in enzymatic tissue dissociation. Collagenase initiates the degradation of the extracellular matrix that in turn leads to loosening of the matrix, exposing protease sensitive sites on other matrix proteins. Purified collagenase can degrade completely purified collagen fibrils or fibers, but digestion of the extracellular matrix requires neutral protease activity to accelerate collagen degradation as well as other extracellular matrix proteins that directly or indirectly hold cells to the matrix. Once a sufficient number of these “anchoring” proteins are cut, then the cells can be released from tissue.
McShane et al. [19] initially showed the importance of neutral protease when they used 18 different lots of crude or enriched collagenase products from Sigma for human islet isolation and categorized these lots for their ability to recover human islets. The lots were scored from 1 to 5 where 1 was “poor” and 5 was “very good.” After further analysis of the enzyme activities reported on the Certificate of Analysis, they selected lots that had similar collagen degradation activities as detected by the Mandl assay but had different neutral protease activities. Those with higher neutral protease activities were effective in recovering human islets where the converse was not true. In 6 islet isolations, they found that supplementing poorly performing lots of collagenase with Dispase led to a significant increase in islet yield. The addition of Dispase alone did not digest the pancreas. Supplementation of poor lots with trypsin or papain led to no improvement of islet yield.
The validity of this observation was confirmed in a subsequent report comparing the effectiveness of Nordmark/Serva, Roche, and VitaCyte purified collagenase and protease enzymes to recover human islets. This report summarized results from 90 human islet isolations using eight different enzyme mixtures [66]. The NEM composed of intact C1 and C2 (VitaCyte CIzyme Collagenase HA) mixed with CHNP (Nordmark/Serva NB Protease) gave significantly shorter digestion times and higher post purification islet yields (n=12) when compared to those isolations using Nordmark/Serva GMP grade enzymes (n=20) or Nordmark/Serva Premium Grade enzymes (n=5).
Further experiments compared the effectiveness of CHNP or thermolysin mixed with a fixed dose of purified collagenase containing intact C1 and C2, to recover islets from a split human pancreas [64]. These results showed that NEM gave significantly higher purified total islet yield and percentage of islets with mean diameters > 200 μm. Analysis of these data expressed as purified IEQ per g pancreas was not significantly different likely because of the small number of experiments performed to compare the performance of these enzymes.
An additional problem arose when Roche changed the extinction coefficient used to estimate the protein concentration of thermolysin in the MTF Kit. Liberase HI and VitaCyte’s Thermolysin product assumed that 1 mg of purified thermolysin had an absorbance of 1.1 at 280 nm (ε0.1% at 280 nm = 1.1). In contrast, the Liberase MTF product used ε0.1% at 280 nm = 1.76 (from Liberase MTF specification) which means that there is at least 60% more enzyme activity per mg of thermolysin than found in Liberase HI. This estimate is consistent with internal studies at VitaCyte that showed the amount of neutral protease activity contained in one 15 mg bottle of thermolysin, a component of the Liberase MTF kit, is equivalent to the activity found in two 12 mg bottles of VitaCyte’s Thermolysin product (unpublished observation). Many laboratories are unaware of this change and continue to assume that thermolysin in the MTF kit is at a similar concentration as Liberase HI (version 4). Others addressed the problem by reducing the amount of thermolysin used in the isolation procedure [6,50].
An alternative to thermolysin or C. histolyticum neutral protease is the P. polymyxa protease (Dispase™ or BP Protease). BP Protease is an animal origin-free product, purified from Paenibacillus polymyxa culture supernatants that do not contain any animal proteins. This protein belongs to the M4 protease family whose members include thermolysin and C. histolyticum neutral protease. Although all members of this family have the same specificity for cleavage at the amino-terminal side of hydrophobic amino acids, they have different selectivity for different amino acids. For example, a study comparing the effectiveness of thermolysin or Dispase to remove epithelial cells from cryopreserved human amniotic membranes showed that each enzyme gave a different degradation pattern of the extracellular matrix proteins associated with this tissue, as detected by immunohistochemical staining methods. Dispase eliminated staining of type VII collagen, laminin 5, and integrins α6 and β4 but retained staining of collagen type IV. By contrast, there was no loss of staining of these proteins after treatment with thermolysin. The relevance of these results shows the ability of similar proteases to digest extracellular matrix proteins differently, depending on the tissue [67]. In contrast, thermolysin, CHNP or BP Protease has been shown to be very effective in recovering islets from human tissue.
Lakey et al. compared the effectiveness of thermolysin and BP Protease to recover human islets when he used Collagenase HA mixed with either 6 mg of VitaCyte’s Thermolysin or 1.1 million neutral protease units (NP U) of BP Protease to digest the pancreas [68]. He performed 12 islet isolations, alternating the enzyme mixtures used for each isolation. Islets were purified over continuous Ficoll gradients, counted, assessed for glucose-stimulated insulin response, and cultured for 48 hours. There were no significant differences in digest time, islet yield per g tissue (3517 ± 156 and 3824 ± 202), and glucose stimulation index (2.4 ± 0.5 and 2.8 ± 0.8) for islets isolated with thermolysin and BP Protease, respectively. However, there was a significant difference (p < 0.05) in cell viability after 48 hours of islet culture (74 ± 5% and 86 ± 2% for thermolysin and BP Protease, respectively) [68].
Further studies by Balamurugan et al. confirmed Lakey et al. observation when they isolated human islets with either purified natural or recombinant C. histolyticum collagenase mixed with 23,400 NP U of BP Protease [54,64]. In these isolations, the body-tail or the entire pancreas was used to recover islets. Data from 18 human islet isolations are shown in Figure 6, where the mean ± one standard deviation for islet recovery was 5,316 ± 818 IEQ/g pancreas under conditions where the purified natural or recombinant collagenase was in excess. Only a limited number of isolations compared the performance of recombinant collagenase– BP Protease enzyme mixtures to the NEM. Here, a higher mean islet yield was obtained with recombinant collagenase-BP Protease mixture, but this difference was not statistically different [64].

Figure 6 Effectiveness of BP Protease for human islet isolation. Box and whiskers plot shows the range (error lines), upper and lower quartile (box), the median (line in the box), and mean (+ in the box). Each point represents results from one isolation. 23,400 NP U of BP Protease/g tissue was added to excess purified natural or recombinant collagenase and used to recover islets from intact pancreata.
A recent report showed intact C1 was more effective in islet recovery than truncated C1 when enzyme mixtures contained C2 but no supplemental protease [69]. In contrast, there was no difference in islet recoveries when the enzyme mixtures above contained supplemental neutral protease activity. The authors suggested that differences in the efficiencies of C1 molecular forms to recover human islets can be overcome if supplemental protease is used in the enzyme mixture. Their conclusion is not supported by the earlier studies cited above where Nordmark/Serva enzymes that contained primarily truncated C1 and neutral protease gave lower islet yields than Collagenase HA–thermolysin or the NEM which contained primarily intact C1 [7,66]. These conclusions may be confounded by clostripain contamination of the Nordmark/Serva collagenase which may activate endogenous pancreatic proteases that increase with cold ischemia time [11,60].
The model presented above serves as a touchstone to formulate a mechanism of how collagenases and proteases degrade the extracellular matrix. As shown above, many different proteases can be used to complement C. histolyticum collagenase for degrading the extracellular matrix. It is assumed that combinations of proteases with different specificities are likely to be more efficient in degradation. Several reports showed improved human islet yields when purified collagenase contaminated with clostripain or when clostripain-thermolysin enzyme mixtures are used in the isolation procedure [49,50]. However, the islet yields from these reports appear to be equivalent or inferior to those that use either the NEM or VitaCyte’s Collagenase HA- BP Protease enzymes [54,66].
The results from experiments using neutral protease enzyme mixtures containing clostripain highlight the limitations of interpreting a “one factor at a time” (OFAT) experimental design to assess the contribution of clostripain to improve human islet yields. An alternative design is to use a DOE approach that enables analysis of two or more factors on measured responses. As noted above, DOE is a more complicated approach than the OFAT experiments but the knowledge gained with this extra effort can be worth the investment.
9. Perspectives and Future Directions for Research
9.1 Perspectives
The model of tissue dissociation presented above serves as a practical guide for selecting enzymes used in the tissue dissociation process. Here, C. histolyticum collagenase plays a very specific role since the digest will not progress until functional collagenase cuts a sufficient number of collagen fibrils or fibers, leading to loosening of the ECM and exposing protease-sensitive sites on ECM proteins. Once the native collagen is cut, neutral protease activity accelerates the degradation of the extracellular matrix. After degradation of a sufficient number of cell anchoring proteins that hold the cells to the ECM, islets are released from the tissue.
Purified collagenase alone is sufficient to degrade native collagen fibrils or fibers. However, collagenase must contain supplemental neutral protease activity to accelerate collagen degradation and degrade other ECM proteins for effective release of islets from the tissue. The ECM is a complex structure where purified collagenase or neutral protease alone are ineffective in releasing islets from tissue. An optimal combination of these enzymes will result in recovery of functional islets.
An interesting implication from this model is that neutral protease is the key enzyme to control when optimizing or validating an enzyme formulation to isolate human islets as long as collagenase CDA is in excess. Excess collagen-degrading activity will have little to no adverse effect on cell isolation because of the narrow selectivity of collagenase to degrade only native collagen or gelatin. The success of this approach is dependent on the characterization of the enzymes used in the isolation process. The collagenase must contain primarily intact enzyme to ensure the highest specific CDA is used for islet isolation. If these criteria are met, then there will be minimal variability between collagenase lots for effective recovery of human islets.
Figure 7 presents an illustrative matrix which summarizes the relationship between neutral protease and collagenase activity and how it impacts cell recovery and viability. This matrix emphasizes the importance of controlling neutral protease activity. The range of optimal activities is likely to be dependent on the dose of a specific protease.

Figure 7 Illustrative matrix showing the effect of the combination of collagenase and neutral protease activities on cell yield and viability
The cost to perform these optimization/validation experiments can be reduced by adopting a human islet isolation method that recovers islets from the head, body, or tail of the same pancreas [70]. This trisected pancreas islet isolation method has recently been used to determine an optimal dose of collagenase and protease for use in isolating human islets from young donors.
The validation experiment requires a fixed dose of intact collagenase at ≥ 20 Wunsch U per g tissue. The mid-range point for neutral protease activity will be either 1.75 DMC U/g tissue for the Nordmark NB Protease, 25,000 NP U/g tissue for VitaCyte’s BP Protease, or 6 mg/g tissue for VitaCyte thermolysin (or 3.8 mg/g tissue of the Roche MTF thermolysin assuming the protein concentration is determined using ε0.1% at 280 nm = 1.76 ). A screening experiment should be performed using a dose of neutral protease activity at 50% or 200% of the midpoint value stated above. The results from two or three isolation should provide a guide to the range of neutral protease activity used in subsequent experiments. These isolations can be performed using intact or trisected pancreas.
There has been long-standing interest to include clostripain in collagenase-protease mixtures used for human islet isolation. Clostripain activity contaminates purified C. histolyticum collagenase or is added as a purified enzyme supplement to purified collagenase-protease enzyme mixtures [49,50]. There are at least two limitations in interpreting the results from these islet isolation experiments. The majority of clostripain is inactive in an oxidizing environment with only about 10% of the enzyme expressing trypsin activity. This activity is reported as “trypsin-like activity” on the certificate of analysis. This activity may fluctuate depending on the oxidation environment in the digest procedure. Moreover, it is difficult to interpret if clostripain is directly or indirectly influencing neutral protease activity. Clostripain’s trypsin-like activity can activate the pancreatic proenzymes, increasing neutral protease activity in the pancreatic tissue digest. Endogenous neutral protease activity dramatically increases immediately before switch time when the closed Ricordi circuit is opened and cells harvested for subsequent processing [71], an observation repeated at VitaCyte during isolation of adult porcine islets (McCarthy, Breite unpublished results). The effect of the addition of clostripain on the kinetics of endogenous neutral protease activity has not been studied to see if there is any correlation between neutral protease activity and islet yield and function.
The addition of multiple proteases with complementary selectivity to excess collagenase CDA is likely to improve islet yield and function for two reasons. It is anticipated that a combination of these enzymes can be used at a lower dose and will release islets at a rapid rate when compared to the use of a single protease. Moreover, recent studies using recombinant C. histolyticum collagenase showed that lower doses of exogenous collagenase increased islet in vitro function without affecting islet yield when compared to islets isolated with higher doses of natural, purified collagenase [54]. The best way to validate this assumption is to use a DOE approach where an excess dose of intact collagenase is fixed with the experimental variables being different doses of neutral proteases with complementary protease activities. The use of the trisected pancreas islet isolation method will reduce the cost of these experiments. The DOE approach should be adopted whenever and wherever possible to ensure that the maximal amount of information is obtained from these expensive and time-consuming cell isolation procedures.
9.2 Future Directions for Research
The information presented above stresses the importance of characterization and definition of the enzyme reagents used in the cell isolation process. The new knowledge on C. histolyticum collagenase structure and function coupled with further studies correlating the specific CDA activity to three functional forms of this enzyme provides a foundation for users to choose the best available collagenase enzymes for human islet isolation. Reports on the three-dimensional structure of collagenase focused primarily on C1 collagenase since there is minimal information on the role C2 collagenase plays in the tissue dissociation process. C2 collagenase has been shown to be more effective in rodent islet isolation than C1 collagenase [72,73] but this same effect was not observed with porcine islet isolation [74]. Moreover, biochemical studies using purified natural C1 and C2 collagenase to degrade collagen fibrils [45] and a recent report using a 30:70 C1:C2 recombinant collagenase-BP Protease enzyme mixtures [54] have shown the effectiveness of C2 in the degradation of collagen fibrils and human islet release, respectively. The elucidation of the role C2 plays in collagen degradation will likely be answered by applying innovative tools, such as atomic force microscopy, to study the role C1 and C2 collagenase play in collagen degradation [55]. This knowledge should provide greater insight into using optimal collagenase enzymes for human islet isolation.
Today’s knowledge of the composition and dose of collagenase-protease enzyme mixtures for human islet isolation is based primarily on donor pancreata recovered from older individuals. Islet recovery from younger donors (< 35 years old) often results in suboptimal islet yields because of the high proportion of embedded and mantled islets [70]. Mantled islets, defined as those islets surrounded by acinar cells, are difficult to purify by density gradient centrifugation because of their different density when compared to non-mantled islets.
As noted above, a recent report used the trisected pancreatic islet isolation model showed that by increasing the dose of neutral protease activity, islet yields from younger donors were comparable to those from older donors. This same method can be applied to improving the recovery of islets from pancreata recovered from chronic pancreatitis patients undergoing total pancreatectomy-autologous islet transplantation to manage their disease. Presently, there are no reports on the development of modified collagenase-protease enzyme formulations to recover human islets from fibrotic pancreata.
The methods to recover human islets should also be re-evaluated since the amount of purified collagenase required to recover human islets from a 100 g pancreas using a modified Ricordi procedure is approximately 5 to 10 times the amount required for digestion of the same mass of human liver tissue used to recover hepatocytes. Comparing results of porcine islet recovery using either a static islet isolation method or the modified Ricordi method, showed no differences in islet yield when the enzyme solution was infused into the main pancreatic duct [75]. There were also no differences in the percentage of digested tissue, the islet size distribution, or the isolation index when these procedures were compared. There were significant differences in the volume of the tissue pellet and the time of the digest (68 ± 2 vs 34 ± 6 min for the static and Ricordi methods, respectively). The decreased tissue pellet volume and longer digestion with the static method suggest that this digestion procedure may provide improved tissue digestion with the smaller pellet volume reflecting less acinar cells in the preparation. In contrast, it is not surprising that the tissue volume is higher when using the Ricordi procedure since the combination of enzymatic and mechanical tissue digestion accelerates the digest process.
A report using a modified static isolation procedure for porcine islet isolation showed the dose of the collagenase-protease enzyme mixtures could be reduced by as much as 67% without any significant difference in islet yield when compared to the control group using the standard dose of enzyme and perfusion volume [76]. Islets were encapsulated into a macrobead and then assessed for basal insulin secretion for 24 hours, once a week during a 12 week period. There was no difference in insulin secretion between islet isolated by the low enzyme versus the standard islet isolation procedure.
The three areas for further study were noted above: the role of C2 in collagen degradation, optimization of enzyme formulations for islet isolations from subsets of pancreata, and re-evaluation of the Ricordi method for islet isolation to determine if islet yield and function could be improved while using lower doses of enzyme. Further studies into these or other areas should focus on improving islet quality as assessed by successful engraftment of an adequate number of human islets required to achieve insulin independence after allo- or auto-islet transplant. Ideally, the correlation of molecular markers or functional assays to estimate the probability of islet engraftment would improve the cost-effectiveness of islet isolation. For this to occur, it will be critical to control the collagenase-protease enzyme mixtures used in the isolation process. The ultimate goal will be to align the biochemical characteristics of these enzyme mixtures to the pancreas characteristics to ensure that the majority of human islet isolations are used in transplants that lead to improved management of adult type 1 diabetic patients.
Acknowledgments
The results from many of the studies summarized in this review used purified enzyme mixtures for VitaCyte LLC. The development of these products was supported by Grant numbers R43DK065467 and R44DK065467 from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). The content is solely the responsibility of the authors and does not necessarily represent the official view of NIDDK or the National Institutes of Health. The authors thank A.N. Balamurugan, A.G. Breite, T.J. Cavanagh, G. Loganathan, S.K. Williams, and M.J. Wright for reviewing the manuscript.
Author Contributions
R.C.M. wrote the review with input from M.L.G. and F.E.D. into the content and final review of the manuscript.
Competing Interests
R.C.M. and F.E.D. are shareholders VitaCyte LLC; R.C.M., F.E.D., and M.L.G. are employees of VitaCyte LLC.
References
- Balamurugan AN, Naziruddin B, Lockridge A, Tiwari M, Loganathan G, Takita M, et al. Islet product characteristics and factors related to successful human islet transplantation from the Collaborative Islet Transplant Registry (CITR) 1999-2010. Amer J Transplant. 2014; 14: 2595-2606. [CrossRef] [Google scholar] [PubMed]
- Shapiro AM, Lakey JR, Ryan EA, Korbutt GS, Toth E, Warnock GL, et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med. 2000; 343: 230-238. [CrossRef] [Google scholar] [PubMed]
- Clinical Islet Transplantation Consortium 2018 [Available from: http://www.citisletstudy.org/.]
- ElSaadany S. Preliminary quantitative risk assessment of developing variant Creutzfeldt-Jakob Disease (vCJD) from human islet transplantation. 2008 [Available from: http://www.med.uottawa.ca/sites/tsalem/documents/Islet%20Risk%20Assessment%20-%20Feb%2018,2008%20-%20final.pdf].
- Brandhorst H, Friberg A, Nilsson B, Andersson HH, Felldin M, Foss A, et al. Large-scale comparison of Liberase HI and collagenase NB1 utilized for human islet isolation. Cell Transplant. 2010; 19: 3-8. [CrossRef] [Google scholar] [PubMed]
- O’Gorman D, Kin T, Imes S, Pawlick R, Senior P, Shapiro AM. Comparison of human islet isolation outcomes using a new mammalian tissue-free enzyme versus Collagenase NB-1. Transplantation. 2010; 90: 255-259. [CrossRef] [Google scholar] [PubMed]
- Balamurugan AN, Breite AG, Anazawa T, Loganathan G, Wilhelm JJ, Papas KK, et al. Successful human islet isolation and transplantation indicating the importance of class 1 collagenase and collagen degradation activity assay. Transplantation. 2010; 89: 954-961. [CrossRef] [Google scholar] [PubMed]
- Ricordi C, Goldstein JS, Balamurugan AN, Szot GL, Kin T, Liu C, et al. National institutes of health-sponsored clinical islet transplantation consortium phase 3 trial: Manufacture of a complex cellular product at eight processing facilities. Diabetes. 2016; 65: 3418-3428. [CrossRef] [Google scholar] [PubMed]
- Paget M, Murray H, Bailey CJ, Downing R. Human islet isolation: semi-automated and manual methods. Diab Vasc Dis Res. 2007; 4: 7-12. [CrossRef] [Google scholar] [PubMed]
- Moassesfar S, Masharani U, Frassetto LA, Szot GL, Tavakol M, Stock PG, et al. A comparative analysis of the safety, efficacy, and cost of islet versus pancreas transplantation in nonuremic patients with type 1 diabetes. Amer J Transplant. 2016; 16: 518-526. [CrossRef] [Google scholar] [PubMed]
- McCarthy RC BA, Green ML, Dwulet FE. Tissue dissociation enzymes for isolating human islets for transplantation: Factors to consider in setting enzyme acceptance criteria. Transplantation. 2011; 91: 137-145. [CrossRef] [Google scholar] [PubMed]
- Brandhorst D, Brandhorst H, Johnson PRV. Enzyme development for human islet isolation: Five decades of progress or stagnation? Review of Diabetic Studies. 2017; 14: 22-38. [CrossRef] [Google scholar] [PubMed]
- Shapiro AMJ, Pokrywczynska M, Ricordi C. Clinical pancreatic islet transplantation. Nat Rev Endocrinol. 2016; 13: 268. [CrossRef] [Google scholar] [PubMed]
- Lakey JR, Burridge PW, Shapiro AM. Technical aspects of islet preparation and transplantation. Transplant Int. 2003; 16: 613-632. [CrossRef] [Google scholar]
- Ricordi C, Tzakis AG, Carroll PB, Zeng YJ, Rilo HL, Alejandro R, et al. Human islet isolation and allotransplantation in 22 consecutive cases. Transplantation. 1992; 53: 407-414. [CrossRef] [Google scholar] [PubMed]
- Cavanagh TJ, Lakey JR, Wright MJ, Fetterhoff T, Wile K. Crude collagenase loses islet-isolating efficacy regardless of storage conditions. Transplant Proc. 1997; 29: 1942-1944. [CrossRef] [Google scholar] [PubMed]
- Hefley TJ, Stern PH, Brand JS. Enzymatic isolation of cells from neonatal calvaria using two purified enzymes from Clostridium histolyticum. Exp Cell Res. 1983; 149: 227-236. [CrossRef] [Google scholar] [PubMed]
- Hatton MW, Berry LR, Krestynski F, Sweeney GD, Regoeczi E. The role of proteolytic enzymes derived from crude bacterial collagenase in the liberation of hepatocytes from rat liver. Identification of two cell-liberating mechanisms. Eur J Biochem. 1983; 137: 311-318. [CrossRef] [Google scholar] [PubMed]
- McShane P, Sutton R, Gray DW, Morris PJ. Protease activity in pancreatic islet isolation by enzymatic digestion. Diabetes. 1989; 38: 126-128. [CrossRef] [Google scholar] [PubMed]
- Fetterhoff TJ, Cavanagh TJ, Wile KJ, Wright MJ, Dwulet FE, Gill J, et al. Human pancreatic dissociation using a purified enzyme blend. Transplant Proc. 1995; 27: 3282-3283. [Google scholar]
- Linetsky E, Bottino R, Lehmann R, Alejandro R, Inverardi L, Ricordi C. Improved human islet isolation using a new enzyme blend, Liberase. Diabetes. 1997; 46: 1120-1123. [CrossRef] [Google scholar] [PubMed]
- Olack BJ, Swanson CJ, Howard TK, Mohanakumar T. Improved method for the isolation and purification of human islets of langerhans using Liberase enzyme blend. Hum Immunol. 1999; 60: 1303-1309. [CrossRef] [Google scholar] [PubMed]
- Wile KJ, Fetterhoff TJ, Cavanagh TJ, Wright MJ. Image analysis of human pancreatic islets: a time-course approach to the evaluation of pancreatic dissociation. Transplant Proc. 1995; 27: 3246-3247. [Google scholar]
- Brandhorst H, Brandhorst D, Hesse F, Ambrosius D, Brendel M, Kawakami Y, et al. Successful human islet isolation utilizing recombinant collagenase. Diabetes. 2003; 52: 1143-1146. [CrossRef] [Google scholar] [PubMed]
- Bucher P, Mathe Z, Morel P, Bosco D, Andres A, Kurfuest M, et al. Assessment of a novel two-component enzyme preparation for human islet isolation and transplantation. Transplantation. 2005; 79: 91-97. [CrossRef] [Google scholar] [PubMed]
- Qi M, Valiente L, McFadden B, Omori K, Bilbao S, Juan J, et al. The Choice of Enzyme for Human Pancreas Digestion is a Critical Factor for Increasing the Success of Islet Isolation. Transplant Dir. 2015; 1: 1-9. [CrossRef] [Google scholar] [PubMed]
- Wünsch E, Heidrich H-G. Zur quantitativen bestimmung der kollagenase. Hoppe-Seyler's Zeitschrift Physiologische Chemie. 1963; 333: 149-151. [CrossRef] [Google scholar] [PubMed]
- Twining SS. Fluorescein isothiocyanate-labeled casein assay for proteolytic enzymes. Anal Biochem. 1984; 143: 30-34. [CrossRef] [Google scholar] [PubMed]
- Mitchell W, Harrington W. Clostripain. Methods Enzymol. 1970; 19: 635-642. [CrossRef] [Google scholar]
- Mandl I, Keller S, Manahan J. Multiplicity of Clostridium histolytcum collagenases. Biochemistry. 1964; 3: 1737-1741. [CrossRef] [Google scholar] [PubMed]
- Bond MD, Van Wart HE. Characterization of the individual collagenases from Clostridium histolyticum. Biochemistry. 1984; 23: 3085-3091. [CrossRef] [Google scholar] [PubMed]
- Hefley TJ. Utilization of FPLC-purified bacterial collagenase for the isolation of cells from bone. J Bone and Mineral Res. 1987; 2: 505-516. [CrossRef] [Google scholar] [PubMed]
- Mitchell WM. The contamination of purified collagenase preparations by clostridiopeptidase B (clostripain): the potential effect on studies utilizing collagenase as a highly specific structural tool. John Hopkins Med J. 1970; 127: 192-198. [Google scholar]
- Dwulet FE, Ellis BB, Gill JF, Jacobsen LB, Smith ME, Waters DG, inventorsPurified mixture of collagenase I, collagenase II, and two other proteases patent. 1998. US Patent # 5, 753, 485. [Google scholar]
- Shapiro AM, Ricordi C, Hering BJ, Auchincloss H, Lindblad R, Robertson RP, et al. International trial of the Edmonton protocol for islet transplantation. N Engl J Med. 2006; 355: 1318-1330. [CrossRef] [Google scholar] [PubMed]
- Lakey JR. Investigating the standardization and effective use of Roche Liberase collagenase blends. 2005. Available from http://icrcohorg/docs/PDF%20Powerpoint%20Conversions/Lakey%20Collagenase%20Slidespdf.
- Barnett MJ, Zhai X, LeGatt DF, Cheng SB, Shapiro AMJ, Lakey JRT. Quantitative assessment of collagenase blends for human islet isolation. Transplantation. 2005; 80: 723-728. [CrossRef] [Google scholar] [PubMed]
- Yamamoto T, Ricordi C, Messinger S, Sakuma Y, Miki A, Rodriguez R, et al. Deterioration and variability of highly purified collagenase blends used in clinical islet isolation. Transplantation. 2007; 84: 997-1002. [CrossRef] [Google scholar] [PubMed]
- Cavanagh TJ, Lakey JR, Wright MJ, Albertson T, Wile K, Fetterhoff TJ. Identification of a pig strain with maximal islet mass. Transplant Proc. 1998; 30: 368. [CrossRef] [Google scholar] [PubMed]
- Bond MD, Van Wart HE. Relationship between the individual collagenases of Clostridium histolyticum: evidence for evolution by gene duplication. Biochemistry. 1984; 23: 3092-3099. [CrossRef] [Google scholar] [PubMed]
- McCarthy RC, Spurlin B, Wright MJ, Breite AG, Sturdevant LK, Dwulet CS, et al. Development and characterization of a collagen degradation assay to assess purified collagenase used in islet isolation. Transplant Proc. 2008; 40: 339-342. [CrossRef] [Google scholar] [PubMed]
- Birk DE, Bruckner P. Collagen suprastructures. Top Curr Chem. 2005; 247: 185-205. [CrossRef] [Google scholar]
- Matsushita O, Okabe A. Clostridial hydrolytic enzymes degrading extracellular components. Toxicon. 2001; 39: 1769-1780. [CrossRef] [Google scholar] [PubMed]
- Bauer R, Janowska K, Taylor K, Jordan B, Gann S, Janowski T, et al. Structures of three polycystic kidney disease-like domains from Clostridium histolyticum collagenases ColG and ColH. Acta Crystallogr Section D, Biol Crystallogr. 2015; 71: 565-577. [CrossRef] [Google scholar] [PubMed]
- Breite AG, McCarthy RC, Dwulet FE. Characterization and functional assessment of Clostridium histolyticum class I (C1) collagenases and the synergistic degradation of native collagen in enzyme mixtures containing class II (C2) collagenase. Transplant Proc. 2011; 43: 3171-3175. [CrossRef] [Google scholar] [PubMed]
- PDB R. Image from the RCSB PDB (www.rcsb.org) of PDB ID 1NQJ (Wilson JJ, Matsushita O, Okabe A, et al.) (2003) A bacterial collagen-binding domain with novel calcium-binding motif controls domain orientation Embo J 22: 1743-1752. www.rcsb.org2003. [CrossRef] [Google scholar] [PubMed]
- PDB R. Image from RCSB PDB (www.rcsb.org) of PDB ID 4TN9 (Bauer R, Janowska K, Taylor K, et al.) 2015) Structures of three polycystic kidney disease-like domains from Clostridium histolyticum collagenase ColG and ColH. Acta Crystallogra Sci Sect D. 2015; 71: 565-577. [CrossRef] [Google scholar] [PubMed]
- PDB R. Image from the RCSB PDB (www.rcsb.org) of PDB ID 4ARE (Eckhard U, Schonauer E, Brandstetter H) (2013) Structural basis for activity regulation and substrate reference of Clostridium collagenases G, H, and T. J Biol Chem 2013;288:20184. [CrossRef] [Google scholar] [PubMed]
- Brandhorst H, Friberg A, Andersson HH, Felldin M, Foss A, Salmela K, et al. The importance of tryptic-like activity in purified enzyme blends for efficient islet isolation. Transplantation. 2009; 87: 370-375. [CrossRef] [Google scholar] [PubMed]
- Stahle M, Foss A, Gustafsson B, Lempinen M, Lundgren T, Rafael E, et al. Clostripain, the Missing Link in the Enzyme Blend for Efficient Human Islet Isolation. Transplant Dir. 2015; 1: e19. [CrossRef] [Google scholar] [PubMed]
- Green ML, Breite AG, Beechler CA, Dwulet FE, McCarthy RC. Effectiveness of different molecular forms of C. histolyticum class I collagenase to recover islets. Islets. 2017: e1365996. PubMed PMID: 28933628. [CrossRef] [Google scholar] [PubMed]
- Kin T, Zhai X, Murdoch TB, Salam A, Shapiro AM, Lakey JR. Enhancing the success of human islet isolation through optimization and characterization of pancreas dissociation enzyme. Amer J Transplant. 2007; 7: 1233-1241. [CrossRef] [Google scholar] [PubMed]
- Frantz C, Stewart KM, Weaver VM. The extracellular matrix at a glance. J Cell Sci. 2010; 123: 4195-4200. [CrossRef] [Google scholar] [PubMed]
- Loganathan G, Subhashree V, Breite AG, Tucker WW, Narayanan S, Dhanasekaran M, et al. Beneficial effect of recombinant rC1rC2 collagenases on human islet function: Efficacy of low-dose enzymes on pancreas digestion and yield. Amer J Transplant. 2018; 18: 478-485. [CrossRef] [Google scholar] [PubMed]
- Watanabe-Nakayama T, Itami M, Kodera N, Ando T, Konno H. High-speed atomic force microscopy reveals strongly polarized movement of clostridial collagenase along collagen fibrils. Scientific Rep. 2016; 6: 28975. PubMed PMID: 27373458. [CrossRef] [Google scholar] [PubMed]
- Karsdal MA, Leeming DJ, Henriksen K, Bay-Jensen A-C. Biochemistry of Collagenase, Laminins, and Elastin. Structure, Function, and Biomarkers. New York: Academic Press; 2016. 238 p. [Google scholar]
- Cross SE, Hughes SJ, Partridge CJ, Clark A, Gray DW, Johnson PR. Collagenase penetrates human pancreatic islets following standard intraductal administration. Transplantation. 2008; 86: 907-911. [CrossRef] [Google scholar] [PubMed]
- Lakey JR, Helms LM, Kin T, Korbutt GS, Rajotte RV, Shapiro AM, et al. Serine-protease inhibition during islet isolation increases islet yield from human pancreases with prolonged ischemia. Transplantation. 2001; 72: 565-570. [CrossRef] [Google scholar] [PubMed]
- AG Breite FE Dwulet, RC McCarthy. Purification and characterization of Clostridium histolyticum neutral protease used for human islet isolation. Transplantation. 2010; 90: 1011. [CrossRef] [Google scholar]
- Kin T, O'Gorman D, Zhai X, Pawlick R, Imes S, Senior P, et al. Nonsimultaneous administration of pancreas dissociation enzymes during islet isolation. Transplantation. 2009; 87: 1700-1705. [CrossRef] [Google scholar] [PubMed]
- Czitrom V. One-factor-at-a-time versus desingned experiments. Am Stat. 1999; 53: 126-131. [Google scholar]
- Wright MJ, Cavanagh TJ, Dwulet FE, Fetterhoff TJ, Smith ME, Wile KJ. Pancreatic dissociation for islet isolation: Development of an optimal enzyme blend through predictive modeling. Miami Bio/Technology Short Reports. 1996; 7: 41-42. [Google scholar]
- Wile KJ, Schwartzkopf W, Olsen M, Laxer C, Fetterhoff T, Cavanagh T, et al. Differentiation of free and embedded porcine pancreatic islets using a novel automated image analysis algorithm. Transplant Proc. 1997; 29: 1974. [CrossRef] [Google scholar] [PubMed]
- Balamurugan AN, Green ML, Breite AG, Loganathan G, Wilhelm JJ, Tweed B, et al. Identifying effective enzyme activity targets for recombinant class I and class II collagenase for successful human islet isolation. Transplant Dir. 2016; 2: e54. PubMed PMID: 27500247. [CrossRef] [Google scholar] [PubMed]
- Matsushita O, Jung CM, Katayama S, Minami J, Takahashi Y, Okabe A. Gene duplication and multiplicity of collagenases in Clostridium histolyticum. J Bacteriol. 1999; 181: 923-933. [Google scholar]
- Balamurugan AN, Loganathan G, Bellin MD, Wilhelm JJ, Harmon J, Anazawa T, et al. A new enzyme mixture to increase the yield and transplant rate of autologous and allogeneic human islet products. Transplantation. 2012; 93: 693-702. [CrossRef] [Google scholar] [PubMed]
- Hopkinson A, Shanmuganathan VA, Gray T, Yeung AM, Lowe J, James DK, et al. Optimization of amniotic membrane (AM) denuding for tissue engineering. Tissue Eng Part C, Methods. 2008; 14: 371-381. [CrossRef] [Google scholar] [PubMed]
- Lakey JRT LM, Li S, Chapman D, Krishnan R, Alexander M, Ichii H, Foster C, Imagawa D, McCarthy R. Replacement of thermolysin in human islet isolation using Bacillus polymyxa protease (Dispase). Joint Congress Cell Transplant Society, International Xenotransplantation Association; (October 23-26 2011). Miami FL. [Google scholar]
- Brandhorst H, Kurfurst M, Johnson PR, Korsgren O, Brandhorst D. Comparison of neutral proteases and collagenase class I as essential enzymes for human islet isolation. Transplant Dir. 2016; 2: e47. PubMed PMID: 27500241. [Google scholar]
- Loganathan G, Subhashree V, Narayanan S, Tweed B, Goedde1 MA, Gunaratnam B, et al. Improved recovery of human islets from young donor pancreases utilizing increased protease dose to collagenase for digesting peri-islet extracellular matrix. Amer J Transplant. 2018; PubMed PMID 30203908 [Google scholar]
- Rose NL, Palcic MM, Helms LM, Lakey JR. Evaluation of Pefabloc as a serine protease inhibitor during human-islet isolation. Transplantation. 2003; 75: 462-466. [CrossRef] [Google scholar] [PubMed]
- Wolters GH, Vos-Scheperkeuter GH, Lin HC, van Schilfgaarde R. Different roles of class I and class II Clostridium histolyticum collagenase in rat pancreatic islet isolation. Diabetes. 1995; 44: 227-233. [CrossRef] [Google scholar] [PubMed]
- Fujio A, Murayama K, Yamagata Y, Watanabe K, Imura T, Inagaki A, et al. Collagenase H is crucial for isolation of rat pancreatic islets. Cell Transplant. 2014; 23: 1187-1198. [CrossRef] [Google scholar] [PubMed]
- Goto M, Yamagata Y, Murayama K, Watanabe K, Imura T, Igarashi Y, et al. Distinct role of collagenase subtypes in the isolation of large animal pancreatic islets. Transplantation. 2013; 96: S142. [Google scholar]
- Dufrane D, D'Hoore W, Goebbels RM, Saliez A, Guiot Y, Gianello P. Parameters favouring successful adult pig islet isolations for xenotransplantation in pig-to-primate models. Xenotransplantation. 2006; 13: 204-214. [CrossRef] [Google scholar] [PubMed]
- Holdcraft RW, Green ML, Breite AG, Circle L, Meyer ED, Adkins H, et al. Optimizing porcine islet isolation to markedly reduce enzyme consumption without sacrificing islet yield or function. Transplant Dir. 2016; 2: e86. PubMed PMID: 27830180. [CrossRef] [Google scholar] [PubMed]



