Chemical Strategies for Improving Islet Transplant Outcomes
Jeremy M. Quintana ![]() , Alexandra L. Stinchcomb
, Alexandra L. Stinchcomb ![]() , Jessica H. Kostyo
, Jessica H. Kostyo ![]() , Blair M. Robichaud
, Blair M. Robichaud ![]() , Michael A. Plunk
, Michael A. Plunk ![]() , Robert R. Kane *
, Robert R. Kane * ![]()
- Department of Chemistry & Biochemistry, Baylor University, 101 Bagby Ave, Waco, TX, USA
* Correspondence: Robert R. Kane ![]()
Academic Editor: Yasuhiko Sugawara
Special Issue: Current Advancement of Islet Cell Transplantation in the Treatment of Diabetes Mellitus
Received: October 8, 2018 | Accepted: December 25, 2018 | Published: December 28, 2018
OBM Transplantation 2018, Volume 2, Issue 4 doi:10.21926/obm.transplant.1804036
Recommended citation: Quintana JM, Stinchcomb AL, Kostyo JH, Robichaud BM, Plunk MA, Kane RR. Chemical Strategies for Improving Islet Transplant Outcomes. OBM Transplantation 2018; 2(4): 036; doi:10.21926/obm.transplant.1804036.
© 2018 by the authors. This is an open access article distributed under the conditions of the Creative Commons by Attribution License, which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is correctly cited.
Abstract
Islet transplantation has proven to be a viable treatment for individuals suffering from both Type 1 Diabetes Mellitus (T1D) and chronic pancreatitis. However, a variety of challenges limit the effectiveness of this procedure by reducing the number of islets that survive the harvesting and transplantation processes. Increasing islet survival would increase the long-term effectiveness of the procedure and allow this technique to be used in more patients. A number of factors have been shown to improve the outcomes of pancreatic islet transplantation, including immunological suppression and prevention of coagulation. In this review, we will discuss various chemical strategies reported in the literature for the preservation of islet graft function after transplantation, including islet encapsulation, the adoption of anti-inflammatory and immune suppressing molecules, and islet surface modification.
Keywords
Islet; allotransplantation; bioconjugation; immunomodulation
1. Introduction
The transplantation of pancreatic islets of Langerhans is an exciting approach to the treatment of brittle diabetes as a result of type 1 diabetes mellitus or type 3c diabetes mellitus (also known as pancreatogenic diabetes). Islet transplantation involves the isolation of islets from a donor pancreas followed by transfer of the isolated islets into a compatible compartment in the recipient (Figure 1). In the first successful clinical islet allotransplant, insulin independence was achieved for only one month before the cells were rejected due to inadequate immunosuppression [1]. In the 445 attempted islet allotransplantations that occurred between 1974 and 2000, 11% of recipients maintained insulin independence for more than a year [2], showing a clear need for improved procedures. By comparison, islet autotransplantation following surgical pancreatectomy typically has higher rates of sustained insulin independence due to the lack of autoimmunity [3,4]. Since 2000, the Edmonton Protocol has been accepted as the clinical standard after it was reported that 100% of the first seven patients to receive this treatment maintained insulin independence through the first year [5]. This success has been attributed to multiple factors including the use of corticosteroid-free immunosuppression and the delivery of a sufficient number of islets from multiple donors. As of September 30, 2015, the Collaborative Islet Transplant Registry had 1,086 allograft and 827 autologous islet transplant recipients registered internationally, with a 7% increase in the number of recipients since the 2013 [6]. In Canada, Australia, Japan, the United Kingdom, France, and other parts of Europe, islet allotransplantation is currently a ‘non-research’ therapeutic option, while in the United States it is an experimental therapy, with clinical trial results recently published by the Consortium in Islet Transplantation in pursuit of a Food and Drug Administration Biological License [7].
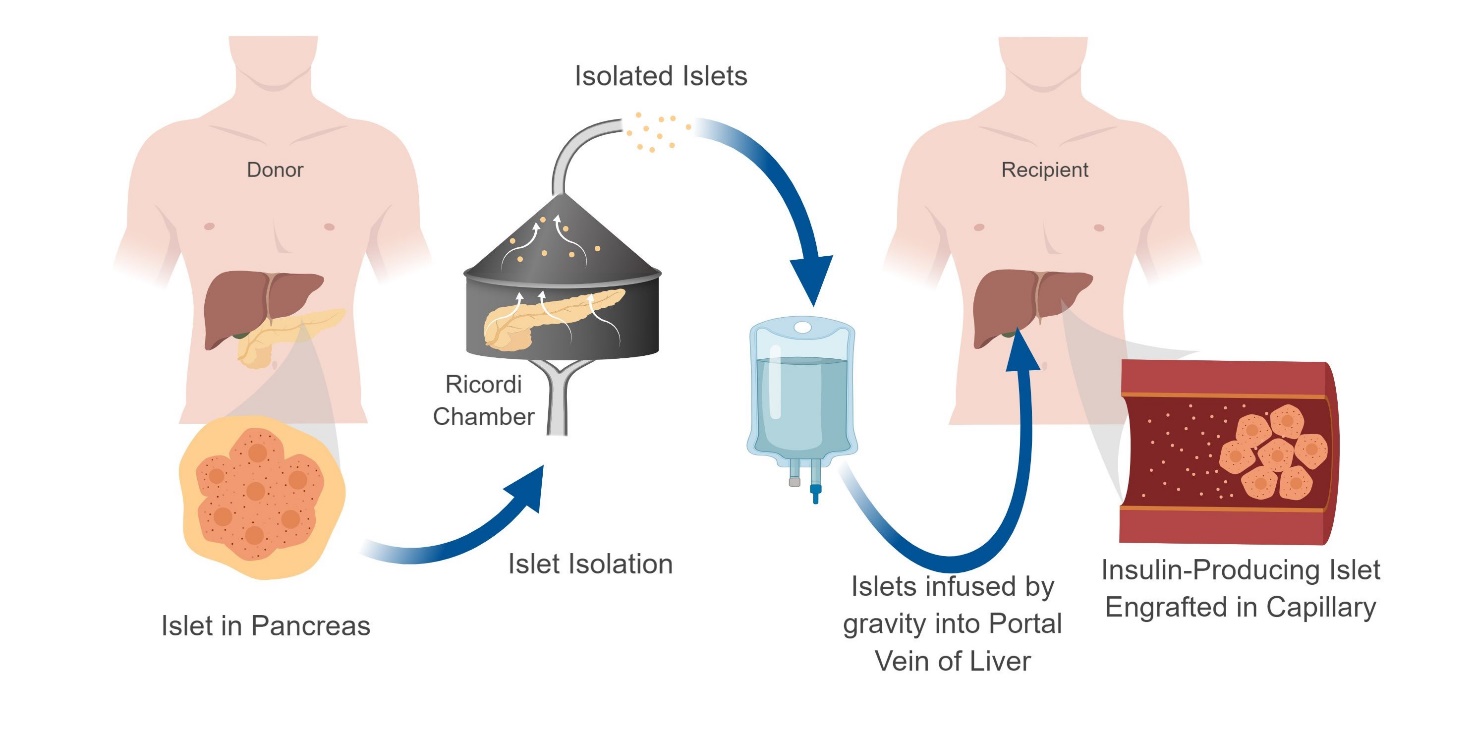
Figure 1 General procedure of intrahepatic islet transplantation.
Although islet transplantation can be successful in returning the patients to normoglycemia, as many as half of the islets die within the first 48 hours after transplantation [8,9]. As a result of this extensive islet death, multiple donors are often required to achieve insulin independence in a recipient, with an average since 2002 of 2.4 donors per recipient [6]. Improvements in islet survival, short-term as well as long-term, will make this life-changing treatment available to more patients worldwide.
A primary factor of islet death is the instant blood-mediated inflammatory response (IBMIR), which involves multiple pathways that results in the destruction of islet allografts, including complement activation and blood clot formation surrounding the islets as well as innate inflammation [10,11,12,13,14,15]. This inflammatory response was originally identified through the use of an assay that detected a thrombotic reaction between human islets and heterologous, ABO-compatible blood [16]. Further investigation of this response showed that islets naturally express tissue factor, which initiates coagulation and the formation of a fibrin capsule around the islet [11,17,18,19]. Platelet activation is one of the primary contributing factors in the generation of an acute inflammatory response, which is followed by macrophage-mediated islet destruction and enhancement of alloantigen-driven immunological attack [17,20]. Similarly, significant numbers of grafted cells undergo apoptosis during the days or weeks following portal infusion [21,22], which may lead to further immune activation.
Another important factor in islet destruction is hypoxia, which is experienced by the islets during isolation, culturing, and after implantation [23,33]. Studies have shown that insufficient oxygen levels lead to cell apoptosis, especially in highly metabolic cells such as β-cells [24,25,26,27,28]. The islet environment within an intact pancreas allows the islets to be highly perfused. For example, in rabbits, islets are located no more than one cell away from capillaries directly leading to arterial circulation [29]. This close proximity to blood flow is necessary for a rapid islet response to glycemic levels, but the consequence is that β-cells only survive optimally when they are exposed to highly oxygenated blood [30,31]. Unfortunately, during isolation and purification the islet capillary network is destroyed [32,33]. Revascularization occurs 10-14 days after intraportal transplantation, when the islets are embolized, settling into the hepatic presinusoidal capillaries [34].
In addition to the complication of maintaining oxygen delivery to transplanted islets, the islets require protection from reactive oxygen species (ROS). These species include nitric oxide, superoxide, peroxynitrite and hydrogen peroxide [35]. Islets are known to have especially high sensitivity toward ROS due to unusually low levels of several antioxidant enzymes, such as superoxide dismutase, glutathione peroxidase, and catalase [36,37]. Although the complete impact of ROS has not been extensively investigated in regard to islet transplantation, it is suspected that reperfusion injury may play a major role in islet graft survival [38,39,40].
New methods to overcome these challenges are currently under investigation, and some of these strategies include the use of a variety of drugs administered directly to the islets pre-transplantation or systemically post-transplantation, in an attempt to address various pathways known to play a part in IBMIR, apoptosis, or coagulation [41]. Alternatively, physical barriers have been employed to prevent the contact of transplanted islet surfaces with immune cells, thus preventing immune activation [42]. These physical barriers include materials for islet encapsulation as well as simple surface modification, and these barriers may also be used in combination with free compounds. Each of these strategies presents unique advantages and disadvantages, and many studies utilize a combination of these approaches. In this review, we will summarize the targets, mechanisms, and advantages and disadvantages of a wide assortment of chemical strategies that have been shown to improve the outcomes of islet transplantation(Table 1).
2. Molecular Therapeutics
Since the introduction of the Edmonton Protocol, pancreatic islet transplantation has become one of the most promising therapeutic strategies for glycemic control in patients with T1D [5,43]. This protocol originally required the infusion of islets within two hours after isolation [44]; however, following the results Froud et al., transplant centers have adopted a period of culturing of the isolated islets prior to transplantation [45]. This culturing allows for quality control testing of the isolated islets, as well as an opportunity to initiate immunosuppression with a variety of drugs, ranging from small, organic molecules (less than 500 Da) to antibodies [46,47]. Transplants performed prior to the implementation of the Edmonton protocol used primarily a combination of azathioprine, cyclosporine, and glucocorticoids, but methods have since evolved to address the limitations of these drugs [5,41].
Current standard protocols for islet transplantation at several transplantation centers focus on preventing activation of T and B cells through inhibition of the mammalian target of rapamycin (mTOR), which is a receptor that plays a role in the regulation of cell growth, cell proliferation, cell motility, cell survival, and protein synthesis [48,49]. The compound rapamycin, also known as sirolimus, has been widely used as a systemic immunosuppressant for use following islet transplantation [50,51,52,53,54]. One of the primary shortcomings of rapamycin is receptor mTORC2 inhibition, which may result in diabetes like symptoms, including decreased glucose tolerance and insensitivity to insulin [55]. Commonly used with sirolimus is tacrolimus [50,53,54,56], also known as FK506, an immunosuppressive drug typically used for allogeneic organ transplant that inhibits the production of interleukin-2 [57], causing suppression of the development and proliferation of T cells and further adaptive immune responses. Unfortunately, this compound has also been found to significantly decrease glucose-induced insulin release even under lower than recommended trough concentrations in murine models [58]. Additionally, when used in combination with sirolimus, even low-dose tacrolimus regiments have been associated with decreased kidney function and kidney failure in some patients [59,60]. Early studies based on the Edmonton protocol also introduced the use of anti-IL-2 antibodies, and two of these antibodies, basiliximab (chimeric) and daclizumab (humanized), have been evaluated in clinical trials [61,62,63,64]. In these trials, the antibodies were well tolerated, and daclizumab was shown to promote a tolerogenic environment promoting graft survival without depletion of Tregs [63].
There has also been interest in preventing downstream activation of the tumor necrosis factor-α (TNF-α) pathway as suggested by Hering et al [65,66]. Inhibiting this pathway in the peritransplant period has been shown to be a promising strategy for facilitating autoimmune regulation and tolerance [7,67,68]. One of the most commonly used TNF-α inhibitors in clinical trials is the biopharmaceutical etanercept, which is typically used in combination with other drugs [69]. Matsumoto et al. reported on the clinical evaluation of combinations of immunosuppressive drugs including etanercept as well as small molecules and antibodies [70]. This study used two different protocols to investigate thymoglobulin induction for improving islet viability by blocking IL-1β and TNF-α. One protocol utilized daclizumab as the induction drug followed by sirolimus and tacrolimus to prolong immunosuppression. The second protocol was a sirolimus-free procedure using a thymoglobulin induction followed by tacrolimus and mycophenolate mofetil, an inhibitor of inosine monophosphate dehydrogenase. The anti-inflammatory drugs used were etanercept, which was used in both protocols, and the anti-IL-1-β antibody, anakinra, which was only used for the second protocol. Islet functionality was determined by both hemoglobin A1c test and the average secretory unit of islet transplant objects index. It was reported that subjects in both protocols were able to achieve euglycemia, although the first method required two islet infusions to achieve the same results as the second method.
Another pathway for immunosuppression in islet transplants is inhibiting the activation of members of the Toll-like Receptor family. These receptors [71] are involved in inflammation via the NF-κB pathway [72]. The TLR family in humans consists of ten transmembrane proteins which act as pattern recognition receptors that bind to specific pathogen-associated molecular patterns (PAMPs) [73]. Some of these receptors also bind endogenous ligands released by damaged cells, known as damage-associated molecular patterns (DAMPs) [74]. TLR4 is the most thoroughly studied member of the TLR family and is most commonly identified as a receptor for bacterial lipopolysaccharide (LPS). However, TLR4 is also a DAMP receptor that has been shown to be activated by high-mobility group box protein 1 (HMGB1), heparan sulfate, hyaluronan fragments, and fibronectin [75]. More recently, TLR4 has been identified as a major contributor to sterile inflammation, acute graft dysfunction and allograft rejection, and autoimmune disease due to its role in the activation of NF-κB [76,77,78,79]. For this reason, several groups have studied the effects of TLR4 inhibition on islet graft survival [80,81].
Dong et al. synthesized a cell-permeable peptide consisting of the protein transfer domain of Hph-1 virus and a fragment of the intracellular domain of TLR4 found in D3h/HeJ mice [82]. It was found that this peptide penetrates cell membranes rapidly, allowing for TLR4 signaling inhibition. The activity of this peptide was studied in the Raw264.7 macrophage line, in isolated islets, and in βTC3 cells. Raw264.7 cells cultured in the presence of the peptide showed lower LPS-induced production of the proinflammatory cytokines IL-1β, TNF-α, iNOS, and IL-6, as well as a reduction in the LPS-induced NF-κB activation. Furthermore, in both isolated islets and β cells the peptide suppressed LPS-induced TNF-α expression through NF-κB inhibition. In murine studies transplant studies it was found that BalB/c (H-2d) islets treated with the peptide had a significantly longer survival rate, with 2 of the 7 grafts surviving past the 100-day mark. The expression of TNF-α and iNOS as well as macrophage infiltration were also reduced by peptide treatment.
Other inhibitors of the NF-κB pathway that have been studied in islet transplantation include Withaferin A, CAY10512, and the TLR4 antagonist TAK-242 [83,84]. Withaferin A inhibits NF-κB kinase beta which prevents phosphorylation of inhibitor of κB (IκB) [85]. Withaferin A and CAY10512, another known NF-κB inhibitor and analog of resveratrol, have been reported to significantly preserve islet viability and decrease tissue factor expression when compared to untreated controls after 48 hours in a miniaturized in vitro tube model of IBMIR [83]. TAK-242, a TLR4 antagonist first reported by Yamada et al. for use as an anti-sepsis agent [86], has more recently been shown to be useful for suppression of the TLR4-stimulated NF-κB pathway in islet transplants [84].
Dexamethasone, an immunosuppressive glucocorticoid, is another small molecule currently being studied for its ability to polarize human blood-derived monocytes toward the anti-inflammatory (M2) phenotype [87,88]. Dexamethasone has also been demonstrated to inhibit the activation of various kinases related to inflammatory gene expression, resulting in suppression of the NF-κB pathway [89]. In mice, this drug has been shown to have a beneficial effect on islet engraftment when incorporated into a polydimethylsiloxane-based, three-dimensional scaffold [90]. The precise dosage is critical when utilizing dexamethasone, as high doses have also been shown to inhibit proper engraftment by preventing healthy host remodeling [91].
Researchers have also targeted pathways of islet apoptosis (Figure 2). The c-Jun NH2-terminal kinases (JNK) are enzymes in the mitogen activated protein kinase (MAPK) family that can be activated by cytokines and environmental stress [92,93]. While the JNK pathway aids in cellular differentiation and proliferation, it also can promote apoptosis by aiding in the transcription of pro-apoptotic genes or activating the caspase signaling cascade leading to mitochondria-mediated cell death [94]. Improper function of the JNK pathway can cause a tolerance towards insulin to develop [95,96,97,98,99,100], and it is believed that the JNK pathway in involved in the onset of type 2 diabetes [95,101]. The JNK pathway is also increasingly activated during the harvest and isolation of islets, peaking after islet digestion and returning to baseline levels after overnight culture [102]. It has been suggested that the JNK pathway causes an aproximate 50% reduction of viable islets during the pancreatic preservation procedure, significantly limiting the number of islets available for transplant post-isolation [103,104,105].
The JNK pathway can be ultimately activated through a signaling cascade originating from various TLR pathways, the most notable being TLR4, as well as by various pro-inflammatory cytokines such as lymphotoxin-β (LT-β), TNF-α, and interleukin 1β (IL-1β) [93]. Several JNK inhibitors (JNKI) have been identified that attenuate islet death during pancreas preservation and islet isolation [106,107]. The peptide 11 arginine JNKI (11R-JNKI), is a cell-permeable JNKI with a chain of 11 arginine that assists with the uptake of the inhibitor into mammalian cells [108]. Noguchi et al. report an improvement of islet graft viability after the introduction of 11R-JNKI during various steps of murine islet transplantation. Transplants of murine islets treated with 11R-JNKI during the preservation, incubation, and transplantation steps resulted in approximately 80% of mice achieving normoglycemia, compared to less than 20% of the three groups not treated with 11RJNKI [105]. Another inhibitor, SP600125, has been shown to inhibit JNK activity via hydrogen bonding at the ATP binding site of JNK [109,110]. Noguchi et al. tested the ability of SP600125 to ameliorate cell death during porcine pancreas isolation. Transplantation of islets isolated from pancreata where SP600125 was introduced into the ductal preservation solution prior to islet isolation resulted in more than 80% of the treatment group achieving normoglycemia, while less than 20% of the non-treated group returned to a normal blood glucose level [102].
Abdelli et al. tested the effects of introducing a peptide consisting of only D-amino acids (D-JNKI) [107]. This inhibitor was injected into the murine pancreatic duct prior to islet isolation, and JNK activity was measured in the isolated islets over the course of two days, after which the islets were stimulated with IL-1β. Kinase assays demonstrated that D-JNKI inhibited JNK activity during the isolation of the islets, and this protection was sustained even after exposure of the islets in culture to IL-1β. D-JNKI also reduced apoptosis in islets exposed to IL-1β [111].
In the early 2000s, Glucagon-like Peptide 1 (GLP-1) was proposed as an antiapoptotic factor and promoter of glucose-dependent insulin production for use in islet transplantation [112]. A study published by Farilla et al. demonstrated the antiapoptotic properties of this peptide in human islets in vitro [113]. In that study, islets were cultured for up to 5 days in the presence of GLP-1 and the expression of caspase-3, B-cell lymphoma-2 (bcl-2), β-actin, and insulin determined via PCR and immunofluorescence. Furthermore, cell apoptosis was quantified through the use of 4’,6-diamidino-phenylindole (DAPI) staining. The GLP-1 treatment afforded a significant reduction in the number of apoptotic cells in GLP-1 treated islets after 3 days, with a 9% decrease in apoptotic nuclei in the treated cultures versus untreated controls. Similarly, the GLP-1 receptor agonist, Exendin-4, has been studied for the enhancement of insulin secretion and overall improvement of clinical outcomes in islet transplantation [114,115,116]. Exendin-4 is an analogue of GLP-1 with a longer half-life, making it more suitable for clinical usage [117]. In a study by Miki et al., this peptide was used in combination with the glucocorticoid methylprednisolone in order to reduce cytokine and chemokine production by human islets in vitro, and produced an 86% increase in the isolated viable beta cell mass versus the untreated control [118].

Figure 2 Biochemical pathways associated with apoptosis including TLR4, TNF-α, IκB, NF-κB, JNK, and caspase.
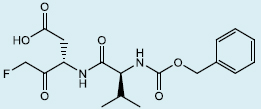
Caspases have also been targeted for inhibition in islet transplantation in order to reduce apoptosis. The majority of post-transplant β-cell apoptosis occurs within the first week following portal infusion, and the caspase family of enzymes is central to this response [21,119]. A number of synthetic peptidyl caspase inhibitors have been explored to limit islet apoptosis, including the pan-caspase inhibitors N-benzyloxycarbonyl-Val-Ala-Asp-fluoromethyl ketone (zVAD-FMK) [120] and N-benzyloxycarbonyl-Val-Asp-fluoromethyl ketone (zVD-FMK) [121], and the caspase-3 specific inhibitor Ac-Asp-Glu-Val-Asp-chloromethylketone (DEVD-CMK) [122]. In murine transplant models using both murine syngeneic islets and human islets, the Shapiro group has shown that zVD-FMK can significantly inhibit engraftment-phase islet cell death and facilitate the use of suboptimal yield islet preparations. Doses as low as 1-3 mg/kg were sufficient to improve the survival of transplanted islets resulting in functional grafts, resulting in the attainment of normoglycemia in recipients even with an 80-90% reduction in the number of transplanted islets [121]. However, the toxicity of some caspase inhibitors may limit the long-term clinical use of these molecules, as their use may lead to increased necrotic cell death [123,124].
Antibodies have also been used to modulate post-transplantation graft survival. In the 2003 paper, Baker et al. [125] utilized an antibody blockade to prevent activation of the chemokine receptor CXCR3 by blocking interaction with the ligand IP-10. Both wild-type islets and CXCR3 knockout islets were prepared and tested in the presence of the inhibitor αIP-10 to determine whether it would delay or prevent rejection by impairing the movement of leukocytes. It was found that treated islets survived longer and had reduced lymphocytic infiltration. Untreated wild-type islets were shown to survive for approximately 13 days, versus the 20 days for both the CXCR3-/- islets and the antibody treated islets. Chemokine expression was statistically similar between the three tested species with both CXCR3-/- and αIP-10 showing 50% reduction of chemokines in the presence of CD8, CD19, and CD4 leukocytes. Other antibodies that have been used for immunosuppression in islet transplantation include the anti-LFA-1 antibody, Efalizumab [126], and the aforementioned anti-IL-1-β antibody, anakinra [69,127], and these antibodies are currently being used in clinical trials [52,56].
The prevention of coagulation is another mechanistic approach to improving islet transplantation success, due to links between coagulation and IBMIR [128]. Antithrombin has been explored for this application [128,129,130]. While alternative thrombin inhibitors such as heparin and hirudin are available, these drugs are limited in terms of efficacy and safety in transplantation [131,132]. Melagatran is an alternative anti-coagulant that was developed to have a high affinity for thrombin with a wide therapeutic window and minimal bleeding complications [133,134]. Özmen et al. have utilized Melagatran in a human islet in vitro model, and these studies demonstrated that thrombin plays an important role in IBMIR and that anti-coagulant drugs like melagatran may be effective in reducing the effects of IBMIR in transplantation [19]. A prodrug for Melagatran, Ximelagatran, was removed from the marked in 2006 due to hepatoxicity [135].
In 2012, Shapiro reported a compilation of seven islet transplant centers worldwide that had achieved a 50% or greater rate of 5-year insulin independence (Table 2). This data was based on a combination of publications, oral presentations, and personal communication and validates the efforts made to improve islet transplant outcomes via the use of molecular therapeutics during the isolation and culturing of islets as well as systemically after transplantation [50].
Table 1 Free compounds that have been used in islet transplantation studies.

Table 2 Centers with five-year insulin independence rates of 50% or greater [50].

3. Islet Encapsulation
Utilization of encapsulation technology to provide a physical barrier between transplanted β-cells and the immune cells of the recipients has provided a promising strategy to reduce or eliminate the need for immunosuppression in islet transplantation [42,153,154]. An ideal encapsulation allows for a normal insulin secretion response to increased blood glucose levels while maintaining cell viability and efficient nutrient and waste exchange (Figure 3). Each encapsulation strategy has unique challenges, such as controlling membrane coating thickness and decreased nutrient and oxygen diffusion [155]. Unfortunately, encapsulation strategies require an increased graft volume, which necessitates the use of larger spaces for the graft, such as the intraperitoneal cavity. In human trials, intraperitoneal infusions of islets have demonstrated microcapsule adhesion to parietal peritoneum, spleen, kidney, and omentum, leading to concerns about the long-term safety of intraperitoneal capsule delivery [156].
Most forms of islet encapsulation are generalized into three categories: macro- ,micro-, and nanoencapsulation, which will be discussed in the next section. The distinction between macro- and microencapsulation is a matter of scale, where macroencapsulation consists of many islets being enclosed within one membrane while microencapsulation consists of a small number, or even individual islets, enclosed within each membrane. These two methods also differ in the surface area to volume ratios, as well as membrane thickness and pore size (Figure 4)[42]. Many methods of encapsulation currently being explored include groups of islets contained within a porous material, which may consist of alginate [157], polyacrylate [158,159], silica [160], collagen [161], chitosan [157,162], gelatin [163], agarose [164], or a combination of these materials. The most significant challenge of encapsulation is finding a material that can simultaneously achieve biocompatibility and immunoisolation, while maintaining islet function. It is also important that the islets retain their relative microenvironment as pancreatic β-cell function is highly dependent on the surrounding matrix environment [165].
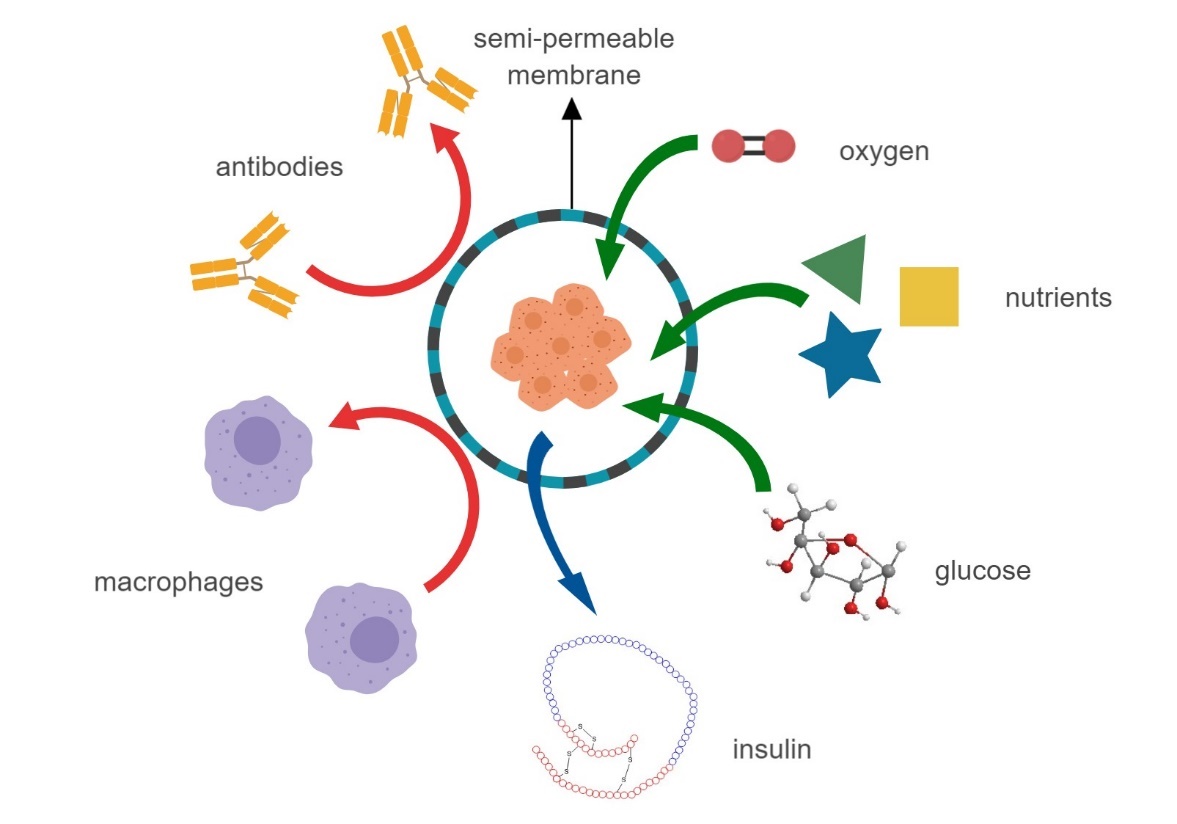
Figure 3 Ideal properties of islet encapsulation.

Figure 4 Visual representation of graft volume versus nutrient diffusion.
The first macroencapsulation strategies, which consisted of transplanted tissue within a porous membrane of either cellulose or nitrocellulose, were first developed by Algire, Prehn, and Weaver in the 1950s [155,166,167]. These encapsulation devices typically had membranes with pore sizes of 450 nm, which was found to be sufficient for preventing direct contact between the transplanted tissue and immune cells. Further studies by Algire and colleagues demonstrated that encapsulation increased graft mass and median graft survival times [168,169]. It was also found that many of these initial experimental transplants eventually failed, despite encapsulation efforts, due to fibroblastic overgrowth of the graft and chamber. Macroencapsulation has been studied using a variety of materials, including polymers such as polytetrafluoroethylene (PTFE) [170] and polycaprolactone (PCL) [171], and inorganic materials such as titania and silicon [172]. Inorganic membranes have the advantages of smaller distribution in pore size and more easily controlled membrane thickness. However, studies have shown that unmodified, rigid materials such as these are more prone to fibrotic encapsulation [173,174], and thus may be more prone to an inflammatory response. In comparison, both PTFE and PCL have demonstrated limited induction of fibrosis, while allowing for revascularization and improved cell viability [170,171].
Alginate encapsulation has been studied as an encapsulation material due to the alginate in vivo biocompatibility, as well as the low cost and ease of production. In the 1980s, the first examples of islet microencapsulation were reported, demonstrating prolonged murine isograft islet survival using alginate-polylysine-polyethyleneimine microcapsules [175]. These early microencapsulation methods showed increased sustainability of normoglycemia in streptozotocin-induced diabetic mice after transplantation into the intraperitoneal space. This strategy was further improved by O’Shea et al. [176], who removed the polyethyleneimine component from the encapsulation material. With this new alginate encapsulation, a murine study was conducted which showed that microencapsulated islets could remain viable for up to 365 days in the intraperitoneal space, as shown by histological studies and glucose challenge. Further efforts to improve the biocompatibility of alginate microcapsules has involved decreasing the impurities and increasing the guluronic acid to mannuronic acid ratio [177,178].
However, variability in alginate production has led to inconsistency in endotoxin content and purity, and, as shown by Rokstad et al. [179], polycation-containing microcapsules, made with materials such as polylysine or poly(methylene-co-guanidine) hydrochloride, may trigger complement and leukocyte activation, while encapsulation with an alginate-divalent cation formula does not. For this reason, the chemical constituents of microcapsules must be closely monitored. Large-scale evaluations, involving thousands of formulations for determining the optimal combinations of polyanions and polycations, have been performed to enable independent control of capsule size, wall thickness, mechanical strength, and permeability. A study conducted by Wang et al. reported over a thousand combinations of polymer constructs and found that a combination of sodium alginate, cellulose sulfate, poly(methylene-co-guanidine) hydrochloride, calcium chloride, and sodium chloride was the most promising for islet protection [180].
Poly(ethylene glycol) (PEG)-based gels, another commonly used encapsulation material, are less immunogenic, non-ionic polymers, and are stable at physiological conditions [181]. Variation in the molecular weight of the PEG polymer can be used to control protein adsorption and permeability of the membrane by adjusting the porosity [182]. One process for the formation of PEG hydrogel is through the use of a photoinitiated, free-radical polymerization, in which the molecular weight of the PEG can affect the cross-link density. This concentration of free radicals is significant for controlling the number of cross-linkable double bonds and for limiting the rate of diffusion across the membrane [183]. In vitro studies by Weber et al. have shown that the insulin secretion observed from murine islets during glucose stimulation was at the same level as unmodified islets when the PEG encapsulation was less than 1 mm in thickness [183,184]. Additionally, Tomei et al. have reported the use of conformal coating to produce a PEG hydrogel coating over islet surfaces [185]. Utilizing a microfluidics technique, conformal coating affords a uniformly thin capsule and minimizes the total graft volume.
In addition to immuno-isolation and prevention of fibrosis, another challenge of islet encapsulation is maintaining adequate oxygen supply for cell functionality [186]. This challenge is especially relevant in the design of macroencapsulation devices, which contain many islets within a robust structure. Studies have shown that insufficient oxygen levels lead to cell apoptosis, especially in highly metabolic cells such as β-cells [24,25]. The delivery of oxygen depends on the diffusion of oxygen from the surrounding blood vessels, to the device, then to the islets within, which requires crossing the immunobarrier membrane. This requirement for proper oxygenation limits the volume of cells that may occupy the device, and thus the geometry of the device. As suggested by the Papas group [187,188], the maximum number of cells is 1,000 IEQ per 1 cm2 surface area for a given encapsulation device. Targeted methods for improved oxygenation include rapid vascularization through the delivery of growth factors [189,190] and the in situ generation of oxygen [191,192]. Early efforts at designing of macroencapsulation devices led to a series of murine transplant studies conducted by Baxter Healthcare, which used a device that contained two membranes sealed at all sides with a loading port [170,193]. This device consisted of PTFE membranes that were specifically designed to encourage host vascularization and allograft immune protection and had pore sizes large enough to allow complete penetration by host cells. The results of these studies demonstrated that vascularization of the membrane-tissue interface was found to be 80-100-fold more effective when the pore size was 5 µm versus when the pore size was 0.02 µm. This significantly higher level of vascularization was maintained for one year in the subcutaneous site in rats.
Localized generation of oxygen has recently been reported through the use of calcium peroxide within polydimethylsiloxane disks [194]. The encapsulation of solid peroxide within a highly hydrophobic material creates a diffusional barrier which can modulate the release of oxygen for more than 40 days. Control of the oxygen release kinetics is achieved through adjustments in the calcium peroxide loading, as well as the size of the disk. Localized delivery of angiogenic factors has also been shown to increase vascularization and enhance islet engraftment and function when utilized with an encapsulation device currently in development by ViaCyte [195]. Interestingly, recruitment of anti-inflammatory monocytes at the site of implantation has also been shown to promote effective vascularization [196].
In order to overcome the obstacle of providing sufficient immunoisolation while providing an oxygen and nutrient-rich environment, Weaver et al. have reported a two-component synthetic PEG hydrogel encapsulation device [197]. This design consists of a hydrogel core that is cross-linked with a non-degradable PEG dithiol and a vasculogenic outer layer cross-linked with a VEGF to promote degradation and enhance localized vascularization. By having a degradable outer layer, this device quickly vascularizes after transplantation to allow for oxygen and nutrient delivery, while the inner layer provides long-term immunoprotection. In a murine model, this macrodevice was implanted in the omentum, and the recipients were monitored for four weeks. After four weeks, the devices were explanted and sectioned to observe the islet viability and functionality, with the results indicating that increased surface vascularization has the capacity to improve encapsulated islet viability.
Materials which are used for encapsulation are also capable carriers for inhibitors of local inflammation [181]. The cytokine transforming growth factor-β (TGFβ) [198] and chemokine CXCL12 [199,200] have been locally delivered in encapsulation materials to suppress the inflammatory response. CXCL12 has been locally delivered by alginate-encapsulated islets to support long-term allogeneic and xenogeneic islet transplantation without systemic immune suppression [201]. This chemokine repels effector T cells while recruiting regulatory T cells (Tregs) and endothelial progenitor cells [202], and serving as a prosurvival signal for beta-cells [203]. Chen et al. have reported that the use of an alginate incorporating CXCL12 as the encapsulation material resulted in a long-term (>300 days) site-specific, immune-protective, and pro-survival environment which protected allo- and xenoislet grafts in murine models [201].
In addition, slow release of TGF-β1 from a poly-lactide-co-glycolide (PLG) islet encapsulation device has been shown to decrease expression of common inflammatory cytokines by 40%, resulting in a 60% decrease in leukocyte infiltration [198].
Interleukin-1 receptor inhibitory peptide (IL-1RIP) has been incorporated into PEG hydrogel capsules as a method of inhibiting the interactions between cytokines and islets. When challenged with pro-inflammatory cytokines, islets which were protected with this peptide-hydrogel showed 75% survival over 21 days while islets that were encapsulated in a standard hydrogel had a survival rate of less than 25% after 4 days [204]. This use of IL-1RIP showed a synergistic protective effect in combination with the hydrogel, and even slightly enhanced the insulin production of the encapsulated islets. It should also be mentioned that the hydrogels used in this study were formed by mixing 4-arm PEG-cysteine macromers and 4-arm PEG with thiol end groups. Thus, the formed hydrogel network exhibited mild reductive properties that provided protection from oxidative stress [205,206].
Of all the encapsulation strategies previously mentioned, a small number have been applied to clinical trials (Table 3). These methods/devices are currently being examined for their safety profiles and efficacy. These strategies include the βAir device developed by Beta-O2, which consists of two major components: an islet module and a gas chamber, which is separated from the islet module. This device is implanted in the subcutaneous tissue, with access ports placed on the dorsal side of the recipient between the scapulae, connected by short polyurethane tubes which allow for daily oxygen filling [207,208]. Also included in the list of devices that are currently undergoing clinical trials are the previously mentioned Sernova Cell Pouch and Encaptra by ViaCyte, which aim to pre-vascularize a subcutaneous site before the addition of islets. Implantation of the Sernova Cell Pouch involves a 2-12 week pre-vascularization period, followed by transplantation of allogeneic islets into channels within the device that are created by removal of a series of rods. The Ecaptra device is an immunoisolating, two-membrane pouch, where the outer membrane has a 5 µm pore size to support cell infiltration and promote angiogenesis. The inner membrane, however, has a pore size of 0.4 µm, which is sufficient for immunoisolation. Another recent clinical trial has evaluated the safety and efficacy of transplanting allogeneic islets encapsulated within a plasma-thrombin scaffold into the omentum [209]. This strategy requires the use of systemic immunosuppression, but does provide the opportunity for successful transplantation into an extrahepatic site and the potential for modulation of the local microenvironment, which may be a crucial foundation for subsequent studies.
Table 3 Islet and β-cell encapsulation systems in recent or current clinical trials.

4. Covalent Surface Modification
A relatively new method for suppressing immunological activation due to islet transplantation involves covalent modification of the islet surface, typically with long chains of polyethylene glycol (PEG) [51,214,215,216,217,218,219,220,221]. This method of protection relies on the flexibility and hydrophilic properties of the PEG chains, which allows for the organization of water molecules to form a hydration shell around the islet and prevent interaction with immunological receptors [222]. Benefits of this anti-inflammatory technique include reduced graft volume, inflammation prevention, and maximum delivery of nutrients for normal islet function. Studies have also shown that surfaces may be modified to tether anticoagulant molecules such as thrombomodulin [223,224] or heparin [225,226], as well as tunable prodrugs which increase islet survivability [227].
The most common approach to islet surface modification is via PEG conjugation using N-hydroxysuccinimide (NHS)-PEG molecules of various lengths [217,218]. The NHS moiety reacts spontaneously under mild conditions with free amines found on the islet surface and has been shown to increase the half-life and reduce immunogenicity of exogenous proteins and blood cells without affecting their function [222]. This same concept has been applied to islet surfaces to mask graft recognition with a simple and efficient conjugation strategy that can be easily performed before transplantation without significant alteration to the transplant procedure. PEGylation alone, however, has not shown significant results, but does show a synergistic effect when used in combination with low-dose immunosuppression [51,216,228].
In a study by Giraldo et al., islet PEGylation in murine subjects was achieved through the exposure of methyl-PEG5000-NHS 48 hours after enzymatic isolation [229]. Groups of mice were then prepared to compare non-PEGylated islets to PEGylated islets in combination with a short-course immunotherapy of anti-LFA-1 antibody for six days. These groups were observed for 100 days to observe the differences in allograft function. It was found that roughly 50% of the recipients of unmodified islets with systemic anti-LFA-1 and those treated with PEGylated islets alone remained normoglycemic for the duration of the study. This was compared to approximately 78% of recipients that remained normoglycemic when treated with a combination of PEGylated islets and immunotherapy. Furthermore, the average times of rejection for the PEGylation alone and immunotherapy alone were 11 days and 25 days respectively, while those who received both treatments had an average rejection time of 35 days. This study demonstrates the effectiveness of a combined approach of PEGylation and immunotherapy.
In 2007 Teramura et al. introduced a new islet surface modification strategy in which Mal-PEG5000-lipids were used to form an ultra-thin membrane of poly(vinyl alcohol) (PVA) anchored to a PEG-phospholipid conjugate [230]. This was done by first reacting Mal-PEG5000-NHS with DPPE phospholipid to synthesize Mal-PEG5000-lipid, which was then reacted with PVA-SH. The resulting PVA-PEG-lipid then introduced to Syrian hamster islets as micelles in aqueous solution. The lipid end of the PEG spacer was then incorporated into the cell membrane of the islets through hydrophobic interactions, while the hydrophilic PEG/PVA region remained outside of the cell membrane. The resulting membrane then underwent autoxidation of the thiol groups to form a multilayered PVA membrane, which did not interfere with the islet’s ability to respond to glucose stimulation. More recently, Haque et al. have reported the use of 6-arm PEG15000 dendrimers to create a multilayered surface modification of porcine islets [51]. This strategy involved the use of unsaturated phospholipids linked to terminal mPEG chains, which modified the islet surface through lipophilic interaction of the phospholipid. When used in combination with an immune suppressant cocktail of tacrolimus, sirolimus, anti-CD154 antibody, and andi-CD19 antibody, this strategy increased the median survival time of porcine islets by 550% in comparison to untreated islets transplanted into murine kidney capsules.
Despite the number of studies conducted involving PEGylation of islets, relatively little has been reported to optimize PEG polymer length used in islet modification. Hashemi et al. has published an extensive comparison of different PEG polymers, showing in a murine model that the incorporation of a variety of polymer lengths was more effective than the use of a single polymer length [216]. The optimal composition for prevention of IL-2 secretion was 75:25% 10 kDa:5 kDa mPEG. Further exploration of PEG length optimization independent of additional immunosuppression would be useful for maximizing the efficiency of islet surface PEGylation.
Aside from amine acylation via NHS-PEG molecules, several other surface bioconjugation strategies have also been shown by SoRelle et al. to successfully modify islet surfaces with biotin without causing significant cell death or inhibiting normal islet function [231]. These strategies include reduction of disulfide bonds followed by Michael additions to maleimide moieties and oxidation of sialic acid using sodium meta-periodate followed by condensation with hydrazide linkers. Surface modification using each of these strategies has been reported in mouse, porcine, and human islets and have resulted in varying amounts and retentions of the modifications. Cell viability in this study was determined by Hoechst 33342/PI stain, and overall modification was determined via detection of biotinylation. The results of these comparisons in bioconjugation strategies across three different islet sources showed relative stability of modification of the human islets in comparison to murine and porcine. Further application of these modification strategies may allow for increased PEGylation density in future transplants, or even allow for multiple protection strategies on a single islet surface.
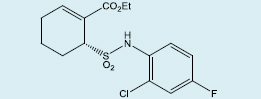
Chang et al. have reported the use of a tunable prodrug which releases the TLR4 antagonist, TAK-242, under physiological conditions to inhibit production of NF-κB in murine studies [227] (Figure 5). The prodrug used in these studies was designed from a prior publication by Santi et al., in which several tunable sulfone linkers were synthesized to demonstrate varying stability based on the pKa of the carbon adjacent to the sulfone modulator [232]. Analysis of the tunable prodrugs from Chang showed a slow, sustained release of the active drug which resulted in a complete blockage of LPS-mediated inflammation in islets as demonstrated by gene and cytokine analysis. Interestingly, this study showed that 100% of the recipients of the modified islets achieved glycemic normalcy even with only half of the standard therapeutic number of islets. This suggests that this method may be used in clinical studies to reduce the dose of islets needed to achieve insulin independence and lead to single-donor transplant success.

Figure 5 Representation of the utilization of immobilized prodrug protection.
Heparin and thrombomodulin have also been covalently attached to the surface of islets to limit coagulation during transplantation. Soluble anticoagulants, such as heparin, have been shown during clinical islet transplantation to only marginally counteract IBMIR [16]. However, Cabric et al. [225] and Jung et al. [233] have reported increased survival of heparin conjugated islets, with Cabric reporting the use of a water-soluble, macromolecular conjugate in which approximately 70 heparin molecules are covalently attached to a polyamine carrier chain via disulfide bonds [234]. This allows for recruitment of plasma proteins and growth factors that have also been found to be beneficial for graft survival, such as vascular endothelial growth factor and fibroblast growth factor [235,236,237]. Bok-Hyeon et al. has reported the effects of heparin modified islets in combination with FK506. The result of this combination of anti-coagulation/anti-inflammatory approaches was a greater than 4-fold increase in mean survival time over unmodified islets in a murine allograft model [226]. Wilson et al. and Stabler et al. reported surface re-engineered pancreatic islets with thrombomodulin and azido-thrombomodulin, respectively, which demonstrated significant increases in islet survivability [223,224]. Wilson reported a threefold increase in the catalytic capacity of surface-bound thrombomodulin to activate the potent anticoagulant, activated protein C, due to a site-specific C-terminal biotinylation strategy. This strategy involved introduction of an azide at the thrombomodulin C-terminus via site-directed mutagenesis, followed by a Stautinger ligation using a triphenylphosphine-PEG3400-biotin conjugate. Surface attachment of anticoagulant molecules offers an alternate method to mimic the protective characteristics of free heparin or thrombomodulin while reducing exposure of collagen and other extracellular matrix proteins, which could potentially reduce or prevent thrombosis and IBMIR.
Other methods for immune suppression include immobilization of ligands to the surface of encapsulation devices. Yolcu et al. have reported the immobilization of a Fas ligand (FasL)-streptavidin chimera to the surface of islets isolated from C57BL/6 mice [238]. This was achieved by first modifying the islet surfaces using sulfo-NHS-LC-biotin, which then bound to the chimera through the biotin-streptavidin interaction. Tolerance for the engrafted islets was maintained by CD4+CD25+Foxp3+ regulatory T cells, resulting in a robust localized tolerance in 100% of C57BL/6 recipients when also treated with a short course of rapamycin. Similarly, Izadi et al. have reported the immobilization of the cell surface ligand jagged-1 (JAG-1) to the surface of murine islets through the use of NHS-PEG3000-Mal crosslinker [239]. Overexpression of JAG-1 in dendritic cells has been shown to induce peripheral tolerance through the support of the differentiation of Tregs [240]. The results of this in vivo study showed a significant increase in the production of anti-inflammatory cytokines IL-10 and TGF-β, while showing a decrease in the pro-inflammatory cytokines IFN-γ and TNF-α.
Hwang et al. have reported the use of a new type of MRI contrast agent with anticoagulant activity via heparin-immobilized superparamagnetic iron oxide (HSPIO) [241]. This HSPIO was chemically conjugated to the islet surface through the use of heterobifunctional PEG linkers, which had both NHS and maleimide moieties on either end of a 3400 Da PEG polymer. The NHS was allowed to react with the islet surface before the maleimide end was reacted with the HSPIO nanoparticles, which contained free thiols. These HSPIO-modified islets were then injected intrahepatically, and in vivo MRI was performed every week for 150 days. Coagulation time increased 6-fold after covalent modification with HSPIO in murine studies. Further experimentation showed that the nanoparticles were rapidly eliminated when not covalently attached, but could provide stable visualization of implanted islets for over 150 days once covalently bound.
5. Conclusions
In the roughly 40 years since islet transplantation was first successfully demonstrated, a number of clear improvements have been made. More than 1,000 patients have had islet transplantations since the introduction of the Edmonton Protocol series in 2000. Prior to that time, 1-year insulin independence rates were below 10% for most islet transplant centres, but these rates have since risen to more than 50% for as long as seven years post-transplant. While multiple transplant centers have achieved insulin independence in patients for more than five-years following islet transplantation, there is a clear need for both alternative islet sources as well as improved efficiency in islet engraftment/survival. A recent review from leading researchers remarks that once the clinical hurdles of “procedural availability, cost and most importantly sustainable reimbursement” are cleared, the development of safer and more reliable methods for generating immunological tolerance can be expected to result in an exponential expansion of the demand for islet transplantation [242].
The critical need for the identification of clinical solutions to T1D, coupled with relatively accessible experimental models of islet transplantation, has resulted in the investigation of a large number of different interventions. Future experimentation will likely see combinations of multiple agents and strategies in order to achieve improved islet transplant effectiveness by affording better control of auto- and alloimmunity. It is also clear that clinical success will likely involve hybrid approaches involving simple biomaterials and/or complex devices, coupled with therapeutics (selected from a wide variety of compounds ranging from small organic drugs to antibodies) targeting multiple pathways. Islet transplantation also provides unique opportunities for the development and evaluation of novel drug delivery strategies, potentially opening the door to the reinvestigation of compounds that may be problematic when dosed systemically. Finally, every one of these strategies will benefit from the development of new islet sources (each of which will present its own unique challenges!). ALL of the approaches described in this review address important aspects of this challenging problem, and as the field evolves there will likely be multiple solutions that incorporate new discoveries coupled with previously described interventions. A recent review by leaders in the field delivers a similar perspective, highlighting the need to address neovascularization, inflammation and oxidative stress, to explore alternative cell sources, to embrace advances in microencapsulation devices, and to adopt effective strategies from recent developments in immune regulation [243].
Importantly, islet transplantation presents a significant opportunity for researchers in a broad array of ‘basic’ sciences to investigate novel pathways and strategies for the protection of transplanted tissue in relevant translational models. This direct link of basic researchers to ‘translational’ research in transplantation is relatively novel, and will ultimately afford improvements in the success of islet transplants while also informing our basic understanding of biological pathways and challenges in transplantation. Ultimately, success in islet transplantation will come from cooperation between a diverse community of clinical and basic researchers and engineers, providing a great incentive for collaboration and cross-disciplinary thinking. This cross-fertilization will undoubtedly have great benefits beyond islet transplantation.
Numerous challenges, including especially the need for multiple donors and life-long, systemic immunosuppression are giving way to new clinical interventions that address the obstacles of IBMIR, apoptosis, and inflammation. Islet transplantation is one of the safest transplant procedures, and overcoming current obstacles will continue to make this an even more attractive therapy. Effective control of auto- and alloimmunity will lead to improved success in single-donor transplants and long-term islet function. Each of the previously discussed methods come with inherent limitations, but increasingly, studies are being published which combine these strategies to mitigate their limitations. Future clinical trials will continue to introduce new islet sources and combinations of islet protection strategies and will further improve the effectiveness of this promising therapy.
Author Contributions
JMQ did literature research, prepared figures, edited coauthor contributions, and was the primary author of the review; ALS did literature research, prepared figures, and edited coauthor contributions; JHK, BMR, and MAP did literature research and prepared preliminary reports; RRK edited the document.
Competing Interests
The authors have declared that no competing interests exist.
References
- Scharp DW, Lacy PE, Santiago JV, McCullough CS, Weide LG, Falqui L, et al. Insulin independence after islet transplantation into type I diabetic patient. Diabetes. 1990; 39: 515518. [CrossRef]
- Brendel MD, Hering BJ, Schultz AO, Bretzel RG. International Islet Transplant Registry. Newsletter #9. 2001; 8. www.med.uni-giessen.de/itr/newsletter/no_9/news_9.html.
- Najarian JS, Sutherland DE, Baumgartner D, Burke B, Rynasiewicz JJ, Matas AJ, et al. Total or near total pancreatectomy and islet autotransplantation for treatment of chronic pancreatitis. Ann Surg. 1980; 192: 526-542. [CrossRef]
- Naziruddin B, Matsumoto S, Noguchi H, Takita M, Shimoda M, Fujita Y, et al. Improved pancreatic islet isolation outcome in autologous transplantation for chronic pancreatitis. Cell Transplant. 2012; 21: 553-558. [CrossRef]
- Shapiro AMJMBBS, Lakey JRTP, Ryan EAMD, Korbutt GSP, Toth EMD, Warnock GLMD, et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoidfree immunosuppressive regimen. N Engl J Med. 2000; 343: 230-238. [CrossRef]
- Eggerman T, Arreaza-Rubin G. CITR Tenth Annual Report. Rockville: Collaborative Islet Transplant Registry, The Emmes Corporation, 2017.
- Hering BJ, Clarke WR, Bridges ND, Eggerman TL, Alejandro R, Bellin MD, et al. Phase 3 trial of transplantation of human islets in type 1 diabetes complicated by severe hypoglycemia. Diabetes Care. 2016; 39: 1230. [CrossRef]
- Toso C, Zaidi H, Morel P, Armanet M, Andres A, Pernin N, et al. Positron-emission tomography imaging of early events after transplantation of islets of langerhans. Transplantation. 2005; 79: 353-355. [CrossRef]
- Eich T, Eriksson O, Sundin A, Estrada S, Brandhorst D, Brandhorst H, et al. Positron emission tomography: A real-time tool to quantify early islet engraftment in a preclinical large animal model. Transplantation. 2007; 84: 893-898. [CrossRef]
- Emamaullee JA, Shapiro AMJ. Factors influencing the loss of β-Cell mass in islet transplantation. Cell Transplant. 2007; 16: 1-8. [CrossRef]
- Johansson H, Lukinius A, Moberg L, Lundgren T, et al. Tissue factor produced by the endocrine cells of the islets of langerhans is associated with a negative outcome of clinical islet transplantation. Diabetes. 2005; 54: 1755-1762. [CrossRef]
- Naziruddin B, Iwahashi S, Kanak MA, Takita M, Itoh T, Levy MF. Evidence for instant bloodmediated inflammatory reaction in clinical autologous islet transplantation. Am J Transplant. 2014; 14: 428-437. [CrossRef]
- Wang J, Sun Z, Gou W, Adams DB, Cui W, Morgan KA, et al. α-1 antitrypsin enhances islet engraftment by suppression of instant blood-mediated inflammatory reaction. Diabetes. 2017; 66: 970-980. [CrossRef]
- Wilson JT, Chaikof EL. Thrombosis and inflammation in intraportal islet transplantation: a review of pathophysiology and emerging therapeutics. J Diabetes Sci Technol. 2008; 2: 746759. [CrossRef]
- Bennet W, Groth C-G, Larsson R, Nilsson B, Korsgren O. Isolated human islets trigger an instant blood mediated inflammatory reaction: implications for intraportal islet transplantation as a treatment for patients with type 1 diabetes. Ups J Med Sci. 2000; 105: 125-133. [CrossRef]
- Bennet W, Sundberg B, Groth C-G, Brendel MD, et al. Incompatibility between human blood and isolated islets of Langerhans: A finding with implications for clinical intraportal islet transplantation?. Diabetes. 1999; 48: 1907-1914. [CrossRef]
- Moberg L, Johansson H, Lukinius A, Berne C, et al. Production of tissue factor by pancreatic islet cells as a trigger of detrimental thrombotic reactions in clinical islet transplantation. Lancet. 2002; 360: 2039-2045. [CrossRef]
- Goto M, Tjernberg J, Dufrane D, Elgue G, Brandhorst D, Ekdahl KN, et al. Dissecting the instant blood-mediated inflammatory reaction in islet xenotransplantation. Xenotransplantation. 2008; 15: 225-234. [CrossRef]
- Özmen L, Ekdahl KN, Elgue G, Larsson R, Korsgren O, Nilsson B. I nhibition of thrombin abrogates the instant blood-mediated inflammatory reaction triggered by isolated human islets: Possible application of the thrombin inhibitor melagatran in clinical islet transplantation. Diabetes. 2002; 51: 1779-1784. [CrossRef]
- Rabinovitch A, Suarez-Pinzon WL. Cytokines and their roles in pancreatic islet beta-cell destruction and insulin-dependent diabetes mellitus. Biochem Pharmacol. 1998; 55: 11391149. [CrossRef]
- Emamaullee J, Liston P, Korneluk RG, Shapiro AMJ, Elliott JF. XIAP Overexpression in Islet βCells Enhances Engraftment and Minimizes Hypoxia–Reperfusion Injury. Am J Transplant. 2005; 5: 1297-1305. [CrossRef]
- Hui H, Khoury N, Zhao X, Balkir L, apos, Amico E, et al. Adenovirus-mediated XIAP gene transfer reverses the negative effects of immunosuppressive drugs on insulin secretion and cell viability of isolated human islets. Diabetes. 2005; 54: 424-433. [CrossRef]
- Delaune V, Berney T, Lacotte S, Toso C. Intraportal islet transplantation: The impact of the liver microenvironment. Transpl Int. 2017; 30: 227-238. [CrossRef]
- Dionne KE, Colton CK, Yarmush ML. Effect of hypoxia on insulin secretion by isolated rat and canine islets of Langerhans. Diabetes. 1993; 42: 12-21. [CrossRef]
- Yoshifumi S, Hiroko E, Hiroaki O, Takaaki T, Hiromi I, Akihisa I, et al. Cellular hypoxia of pancreatic beta-cells due to high levels of oxygen consumption for insulin secretion in vitro. J Biol Chem. 2011; 286: 12524-12532. [CrossRef]
- Duvillie B. Hypoxia and oxidative stress during the transplantation of islets of Langerhans. Correspondances en MHND. 2015; 19: 53-57.
- Hyder A, Laue C, Schrezenmeir J. Variable responses of islet cells of different ages and species to hypoxia 11 supported by the grant of “Stiftung Insel Transplantation” to Ch. Laue and by the Egyptian government grant to A. Hyder. Transplantation Proceedings. 1998; 30: 578-580. [CrossRef]
- Davalli AM, Scaglia L, Zangen DH, Hollister J, Bonner-Weir S, Weir GC. Vulnerability of islets in the immediate posttransplantation period. Dynamic changes in structure and function. Diabetes. 1996; 45: 1161-1167 [CrossRef]
- Lifson N, Kramlinger KG, Mayrand RR, Lender EJ. Blood flow to the rabbit pancreas with special reference to the islets of langerhans. Gastroenterology. 1980; 79: 466-473. [CrossRef]
- Carlsson P-O, Palm F, Andersson A, Liss P. Markedly decreased oxygen tension in transplanted rat pancreatic islets irrespective of the implantation site. Diabetes. 2001; 50: 489-495. [CrossRef]
- Kühtreiber WM, Lanza RP, Beyer AM, Kirkland KS, Chick WL. Relationship between insulin secretion and oxygen tension in hybrid diffusion chambers. ASAIO J. 1993; 39: M247-M251. [CrossRef]
- Menger MD, Jaeger S, Walter P, Feifel G, Hammersen F, Messmer K. Angiogenesis and hemodynamics of microvasculature of transplanted islets of langerhans. Diabetes. 1989; 38: 199-201. [CrossRef]
- Stagner JI, Mokshagundam S, Samols E. Hormone secretion from transplanted islets is dependent upon changes in islet revascularization and islet architecture. Transplant Proc. 1995; 27: 3251-3254.
- Jones GL, Juszczak MT, Hughes SJ, Kooner P, Powis SH, Press M. Time course and quantification of pancreatic islet revasculariztion following intraportal transplantation. Cell Transplant. 2007; 16: 505-516. [CrossRef]
- Becker LB. New concepts in reactive oxygen species and cardiovascular reperfusion physiology. Cardiovasc Res. 2004; 61: 461-470. [CrossRef]
- Lenzen S, Drinkgern J, Tiedge M. Low antioxidant enzyme gene expression in pancreatic islets compared with various other mouse tissues. Free Radic Biol Med. 1996; 20: 463-466. [CrossRef]
- Tiedge M, Lortz S, Drinkgern J, Lenzen S. Relation between antioxidant enzyme gene expression and antioxidative defense status of insulin-producing cells. Diabetes. 1997; 46: 1733-1742. [CrossRef]
- Peltenburg HG, Wolffenbuttel BH, Booster MH, Menheere PP, Leunissen KM, Kootstra G, et al. Reperfusion injury of pancreas allografts: Relation to islet cell function. Transpl Int. 1992; 5: S270-S271. [CrossRef]
- Pitchumoni S, Garfinkel MR, Littman ED, Opara EC. Reoxygenation injury affects isolated islet response to fatty acid stimulation. Metabolism. 1998; 47: 809-813. [CrossRef]
- Vollmar B, Janata J, Yamauchi J, Wolf B, Heuser M, Menger MD. Exocrine, but not endocrine, tissue is susceptible to microvascular ischemia/reperfusion injury following pancreas transplantation in the rat. Transpl Int. 1999; 12: 50-55. [CrossRef]
- Shapiro AMJ, Nanji Sulaiman A, Lakey Jonathan RT. Clinical islet transplant: Current and future directions towards tolerance. Immunol Rev. 2003; 196: 219-236. [CrossRef]
- Desai T, Shea LD. Advances in islet encapsulation technologies. Nat Rev Drug Discov. 2016; 16: 338. [CrossRef]
- Shapiro AMJMDP, Ricordi CMD, Hering BJMD, Auchincloss HMD, Lindblad RMD, Robertson RPMD, et al. International trial of the edmonton protocol for islet transplantation. N Engl J Med. 2006; 355: 1318-1330. [CrossRef]
- Shapiro AMJ, Ricordi C, Hering BJ, Auchincloss H, Lindblad R, Robertson RP, et al. International trial of the edmonton protocol for islet transplantation. N Engl J Med. 2006; 355: 1318-1330. [CrossRef]
- Froud T, Ricordi C, Baidal DA, Hafiz MM, Ponte G, Cure P, et al. Islet transplantation in type 1 diabetes mellitus using cultured islets and steroid-free immunosuppression: Miami experience. Am J Transplant. 2005; 5: 2037-2046. [CrossRef]
- Belle TV, Herrath Mv. Immunosuppression in islet transplantation. J Clin Invest. 2008; 118: 1625-1628.
- Noguchi H, Matsumoto S, Matsushita M, Kobayashi N, Tanaka K, Matsui H, et al. Immunosuppression for islet transplantation. Acta Med Okayama. 2006; 60: 71-76.
- Lipton Jonathan O, Sahin M. The neurology of mtor. Neuron. 2014; 84: 275-291. [CrossRef]
- Nissim Hay NS. Upstream and downstream of mtor. Genes Dev. 2004; 18: 1926-1945. [CrossRef]
- Shapiro AMJ. Islet transplantation in type 1 diabetes: Ongoing challenges, refined procedures, and long-term outcome. Rev Diabet Stud. 2012; 16: 385-406. [CrossRef]
- Haque MR, Jeong J-H, Byun Y. Combination strategy of multi-layered surface camouflage using hyperbranched polyethylene glycol and immunosuppressive drugs for the prevention of immune reactions against transplanted porcine islets. Biomaterials. 2016; 84: 144-156. [CrossRef]
- Posselt AM, Szot GL, Frassetto LA, Masharani U, Tavakol M, Amin R, et al. Islet transplantation in type 1 diabetic patients using calcineurin inhibitor-free immunosuppressive protocols based on t-cell adhesion or costimulation blockade. Transplantation. 2010; 90: 1595-1601. [CrossRef]
- Gangemi A, Salehi P, Hatipoglu B, Martellotto J, Barbaro B, Kuechle JB, et al. Islet transplantation for brittle type 1 diabetes: The uic protocol. Am J Transplant. 2008; 8: 12501261. [CrossRef]
- Lablanche S, Borot S, Wojtusciszyn A, Bayle F, Tetaz R, Badet L, et al. Five-year metabolic, functional, and safety results of patients with type 1 diabetes transplanted with allogenic islets within the swiss-french gragil network. Diabetes Care. 2015; 38: 1714-1722. [CrossRef]
- Lamming DW, Ye L, Katajisto P, Goncalves MD, Saitoh M, Stevens DM, et al. Rapamycininduced insulin resistance is mediated by mtorc2 loss and uncoupled from longevity. Science. 2012; 335: 1638. [CrossRef]
- Senior PA, Kin T, Shapiro J, Koh A. Islet transplantation at the university of alberta: Status update and review of progress over the last decade. Can J Diabetes. 2012; 36: 32-37. [CrossRef]
- Sawada S, Suzuki G, Kawase Y, Takaku F. Novel immunosuppressive agent, fk506. In vitro effects on the cloned t cell activation. J Immunol. 1987; 139: 1797.
- Radu RG, Fujimoto S, Mukai E, Takehiro M, Shimono D, Nabe K, et al. Tacrolimus suppresses glucose-induced insulin release from pancreatic islets by reducing glucokinase activity. Am J Physiol Endocrinol Metab. 2005; 288: E365-E371. [CrossRef]
- Andres A, Toso C, Morel P, Demuylder-Mischler S, Bosco D, Baertschiger R, et al. Impact of a sirolimus/tacrolimus-based immunosuppressive regimen on kidney function after islet transplantation. Transplant Proc. 2005; 37: 1326-1327. [CrossRef]
- Leitão CB, Cure P, Messinger S, Pileggi A, Lenz O, Froud T, et al. Stable renal function after islet transplantation: Importance of patient selection and aggressive clinical management. Transplantation. 2009; 87: 681-688. [CrossRef]
- Rickels MR, Liu C, Shlansky-Goldberg RD, Soleimanpour SA, Vivek K, Kamoun M, et al. Improvement in β-cell secretory capacity after human islet transplantation according to the cit07 protocol. Diabetes. 2013; 62: 2890-2897. [CrossRef]
- Ryan EA, Lakey JRT, Rajotte RV, Korbutt GS, Kin T, Imes S, et al. Clinical outcomes and insulin secretion after islet transplantation with the edmonton protocol. Diabetes. 2001; 50: 710-719. [CrossRef]
- Hire K, Ngo DK, Stewart-Maynard KM, Hering B, Bansal-Pakala P. Foxp3+, and not cd25+, t cells increase post-transplant in islet allotransplant recipients following anti-cd25+ ratg immunotherapy. Cell Immunol. 2012; 274: 83-88. [CrossRef]
- Toso C, Edgar R, Pawlick R, Emamaullee J, Merani S, Dinyari P, et al. Effect of different induction strategies on effector, regulatory and memory lymphocyte sub-populations in clinical islet transplantation. Transpl Int. 2009; 22: 182-191. [CrossRef]
- Hering BJ, Kandaswamy R, Ansite JD, et al. Single-donor, marginal-dose islet transplantation in patients with type 1 diabetes. JAMA. 2005; 293: 830-835. [CrossRef]
- Bellin MD, Barton FB, Heitman A, Harmon JV, Kandaswamy R, Balamurugan AN, et al. Potent induction immunotherapy promotes long-term insulin independence after islet transplantation in type 1 diabetes. Am J Transplant. 2012; 12: 1576-1583. [CrossRef]
- Bellin MD, Kandaswamy R, Parkey J, Zhang HJ, Liu B, Ihm SH, et al. Prolonged insulin independence after islet allotransplants in recipients with type 1 diabetes. Am J Transplant. 2008; 8: 2463-2470. [CrossRef]
- Parker MJ, Xue S, Alexander JJ, Wasserfall CH, Campbell-Thompson ML, Battaglia M, et al. Immune depletion with cellular mobilization imparts immunoregulation and reverses autoimmune diabetes in nonobese diabetic mice. Diabetes. 2009; 58:2277-2284. [CrossRef]
- Takita M, Matsumoto S, Shimoda M, Chujo D, Itoh T, SoRelle JA, et al. Safety and tolerability of the t-cell depletion protocol coupled with anakinra and etanercept for clinical islet cell transplantation. Clin Transplant. 2012; 26: E471-E484. [CrossRef]
- Matsumoto S, Takita M, Chaussabel D, Noguchi H, Shimoda M, Sugimoto K, et al. Improving efficacy of clinical islet transplantation with iodixanol-based islet purification, thymoglobulin induction, and blockage of il-1β and tnf-α. Cell Transplant. 2011; 20: 1641-1647. [CrossRef]
- Martin AG, Fresno M. Tumor necrosis factor-α activation of nf-κb requires the phosphorylation of ser-471 in the transactivation domain of c-rel. J Biol Chem. 2000; 275: 24383-24391. [CrossRef]
- Kawai T, Akira S. Signaling to nf-κb by toll-like receptors. Trends Mol Med. 2007; 13: 460-469. [CrossRef]
- Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006; 124: 783-801. [CrossRef]
- Oliveira JB, Soares AASM, Sposito AC. Chapter two - inflammatory response during myocardial infarction. Adv Clin Chem. 2018; 84: 39-79. [CrossRef]
- Barton GM. A calculated response: Control of inflammation by the innate immune system. J Clin Invest. 2008; 118: 413-420. [CrossRef]
- Krüger B, Yin N, Zhang N, Yadav A, Coward W, Lal G, et al. Islet-expressed tlr2 and tlr4 sense injury and mediate early graft failure after transplantation. Eur J Immunol. 2010; 40: 29142924. [CrossRef]
- Krüger B, Yin N, Zhang N, Yadav A, Coward W, Lal G, et al. Islet-expressed tlr2 and tlr4 sense injury and mediate early graft failure after transplantation. Eur J Immunol. 2010; 40: 29142924. [CrossRef]
- Ro H, Hong J, Kim BS, Lee EW, Kim M-G, Han KH, et al. Roles of toll-like receptors in allogeneic islet transplantation. Transplantation. 2012; 94: 1005-1012. [CrossRef]
- Gao Q, Ma LL, Gao X, Yan W, Williams P, Yin DP. Tlr4 mediates early graft failure after intraportal islet transplantation. Am J Transplant. 2010; 10: 1588-1596. [CrossRef]
- Goldberg A, Parolini M, Chin BY, Czismadia E, Otterbein LE, Bach FH, et al. Toll-like receptor 4 suppression leads to islet allograft survival. FASEB J. 2007; 21: 2840-2848. [CrossRef]
- Wang D, Jiang L, Liang Y, Hao X, Chen C, Xia W, et al. Comp-ang1 promotes long-term survival of allogeneic islet grafts in a bioinert perforated chamber by inhibiting inflammation via inhibition of the tlr4 signaling pathway. Biotechnol Lett. 2016; 38: 1033-1042. [CrossRef]
- Dong H, Zhang Y, Song L, Kim D-S, Wu H, Yang L, et al. Cell-permeable peptide blocks tlr4 signaling and improves islet allograft survival. Cell Transplant. 2016; 25: 1319-1329. [CrossRef]
- Kanak MA, Takita M, Itoh T, SoRelle JA, Murali S, Kunnathodi F, et al. Alleviation of instant blood-mediated inflammatory reaction in autologous conditions through treatment of human islets with nf-κb inhibitors. Transplantation. 2014; 98: 578-584. [CrossRef]
- Chang CA, Murphy K, Kane RR, Lawrence MC, Naziruddin B. Early tlr4 blockade attenuates sterile inflammation-mediated stress in islets during isolation and promotes successful transplant outcomes. Transplantation. 2018; 102: 1505-1513. [CrossRef]
- Grover A, Shandilya A, Punetha A, Bisaria VS, Sundar D. Inhibition of the nemo/ikkβ association complex formation, a novel mechanism associated with the nf-κb activation suppression by withania somnifera’s key metabolite withaferin a. BMC Genomics. 2010; 11: S25. [CrossRef]
- Yamada M, Ichikawa T, Ii M, Itoh K, Tamura N, Kitazaki T. Novel cyclohexene derivatives as anti-sepsis agents: Synthetic studies and inhibition of no and cytokine production. Bioorg Med Chem. 2008; 16: 3941-3958. [CrossRef]
- Tedesco S, Bolego C, Toniolo A, Nassi A, Fadini GP, Locati M, et al. Phenotypic activation and pharmacological outcomes of spontaneously differentiated human monocyte-derived macrophages. Immunobiology. 2015; 220: 545-554. [CrossRef]
- Tuckermann JP, Kleiman A, McPherson KG, Reichardt HM. Molecular mechanisms of glucocorticoids in the control of inflammation and lymphocyte apoptosis. Crit Rev Clin Lab Sci. 2005; 42: 71-104. [CrossRef]
- Moynagh P. Toll-like receptor signalling pathways as key targets for mediating the antiinflammatory and immunosuppressive effects of glucocorticoids. J Endocrinol. 2003; 179: 139-144. [CrossRef]
- Jiang K, Weaver JD, Li Y, Chen X, Liang J, Stabler CL. Local release of dexamethasone from macroporous scaffolds accelerates islet transplant engraftment by promotion of antiinflammatory m2 macrophages. Biomaterials. 2017; 114: 71-81. [CrossRef]
- Franz S, Rammelt S, Scharnweber D, Simon JC. Immune responses to implants – a review of the implications for the design of immunomodulatory biomaterials. Biomaterials. 2011; 32: 6692-6709. [CrossRef]
- Ip YT, Davis RJ. Signal transduction by the c-jun n-terminal kinase (jnk) — from inflammation to development. Curr Opin Cell Biol. 1998; 10: 205-219. [CrossRef]
- Ammendrup A, Maillard A, Nielsen K, Nina Aabenhus A, Serup P, et al. The c-jun aminoterminal kinase pathway is preferentially activated by interleukin-1 and controls apoptosis in differentiating pancreatic beta-cells. Diabetes. 2000; 49: 1468-1420. [CrossRef]
- Dhanasekaran DN, Reddy EP. Jnk signaling in apoptosis. Oncogene. 2008; 27: 6245-6251. [CrossRef]
- Hirosumi J, Tuncman G, Chang L, Gorgun CZ, et al. A central role for jnk in obesity and insulin resistance. Nature. 2002; 420: 333-336. [CrossRef]
- Aguirre V, Uchida T, Yenush L, Davis R, White MF. The c-jun nh(2)-terminal kinase promotes insulin resistance during association with insulin receptor substrate-1 and phosphorylation of ser(307). J Biol Chem. 2000; 275: 9047-9054. [CrossRef]
- Noguchi H, Nakai Y, Ueda M, Masui Y, Futaki S, Kobayashi N, et al. Activation of c-jun nh2terminal kinase (jnk) pathway during islet transplantation and prevention of islet graft loss by intraportal injection of jnk inhibitor. Diabetologia. 2007; 50: 612-619. [CrossRef]
- Kaneto H, Xu G, Fujii N, Kim S, Bonner-Weir S, Weir GC. Involvement of c-jun n-terminal kinase in oxidative stress-mediated suppression of insulin gene expression. J Biol Chem. 2002; 277: 30010-30018. [CrossRef]
- Manning AM, Davis RJ. Targeting jnk for therapeutic benefit: From junk to gold?. Nat Rev Drug Discov. 2003; 2: 554+. [CrossRef]
- Standaert ML, Bandyopadhyay G, Galloway L, Soto J, Ono Y, Kikkawa U, et al. Effects of knockout of the protein kinase c β gene on glucose transport and glucose homeostasis1. Endocrinology. 1999; 140: 4470-4477. [CrossRef]
- Bennett BL, Satoh Y, Lewis AJ. Jnk: A new therapeutic target for diabetes. Curr Opin Cell Biol. 2003; 3: 420-425. [CrossRef]
- Noguchi H, Matsumoto S, Onaca N, Naziruddin B, Jackson A, Ikemoto T, et al. Ductal injection of jnk inhibitors before pancreas preservation prevents islet apoptosis and improves islet graft function. Hum Gene Ther. 2009; 20: 73+. [CrossRef]
- Matsumoto I, Sawada T, Nakano M, Sakai T, Liu B, Ansite JD, et al. Improvement in islet yield from obese donors for human islet transplants. Transplantation. 2004; 78: 880-885. [CrossRef]
- Ichii H, Wang X, Messinger S, Alvarez A, Fraker C, Khan A, et al. Improved human islet isolation using nicotinamide. Am J Transplant. 2006; 6: 2060-2068. [CrossRef]
- Noguchi H, Matsumoto S, Kobayashi N, Hayashi S, Iwanaga Y, Nagata H, et al. Effect of jnk inhibitor during islet isolation and transplantation. Transplant Proc. 2008; 40: 379-381. [CrossRef]
- Noguchi H, Miyagi-Shiohira C, Nakashima Y, Ebi N, Hamada E, Tamaki Y, et al. Modified cellpermeable jnk inhibitors efficiently prevents islet apoptosis and improves the outcome of islet transplantation. Sci Rep. 2018; 8: 1-9. [CrossRef]
- Bonny C, Oberson A, Negri S, Sauser C, Schorderet DF. Cell-permeable peptide inhibitors of jnk. Diabetes. 2001; 50: 77. [CrossRef]
- Noguchi H, Nakai Y, Matsumoto S, Kawaguchi M, Ueda M, Okitsu T, et al. Cell permeable peptide of jnk inhibitor prevents islet apoptosis immediately after isolation and improves islet graft function. Am J Transplant. 2005; 5: 1848-1855. [CrossRef]
- Bennett BL, Sasaki DT, Murray BW, O'Leary EC, Sakata ST, Xu W, et al. Sp600125, an anthrapyrazolone inhibitor of jun n-terminal kinase. Proc Natl Acad Sci U S A. 2001; 98: 13681-13686. [CrossRef]
- Heo Y-S, Kim S-K, Seo CI, Kim YK, Sung B-J, Lee HS, et al. Structural basis for the selective inhibition of jnk1 by the scaffolding protein jip1 and sp600125. EMBO J. 2004; 23: 2185-2195. [CrossRef]
- Abdelli S, Abderrahmani A, J Hering B, Beckmann J, Bonny C. The c-jun n-terminal kinase jnk participates in cytokine- and isolation stress-induced rat pancreatic islet apoptosis. Diabetologia. 2007; 50: 1660-1669. [CrossRef]
- Farilla L, Hui H, Bertolotto C, Kang E, Bulotta A, Di Mario U, et al. Glucagon-like peptide-1 promotes islet cell growth and inhibits apoptosis in zucker diabetic rats. Endocrinology. 2002; 143: 4397-4408. [CrossRef]
- Farilla L, Bulotta A, Hirshberg B, Li Calzi S, Khoury N, Noushmehr H, et al. Glucagon-like peptide 1 inhibits cell apoptosis and improves glucose responsiveness of freshly isolated human islets. Endocrinology. 2003; 144: 5149-5158. [CrossRef]
- Carlessi R, Lemos NE, Dias AL, Oliveira FS, Brondani LA, Canani LH, et al. Exendin-4 protects rat islets against loss of viability and function induced by brain death. Mol Cell Endocrinol. 2015; 412: 239-250. [CrossRef]
- Ghofaili KA, Fung M, Ao Z, Meloche M, Shapiro RJ, Warnock GL, et al. Effect of exenatide on β cell function after islet transplantation in type 1 diabetes. Transplantation. 2007; 83: 24-28. [CrossRef]
- Faradji RN, Tharavanij T, Messinger S, Froud T, Pileggi A, Monroy K, et al. Long-term insulin independence and improvement in insulin secretion after supplemental islet infusion under exenatide and etanercept. Transplantation. 2008; 86: 1658-1665. [CrossRef]
- Parkes D, Jodka C, Smith P, Nayak S, Rinehart L, Gingerich R, et al. Pharmacokinetic actions of exendin-4 in the rat: Comparison with glucagon-like peptide-1. Drug Dev Res. 2001; 53: 260267. [CrossRef]
- Miki A, Ricordi C, Yamamoto T, Sakuma Y, Misawa R, Mita A, et al. Improved human islet preparations using glucocorticoid and exendin-4. Pancreas. 2014; 43: 1317-1322. [CrossRef]
- Emamaullee JA, Rajotte RV, Liston P, Korneluk RG, Lakey JRT, Shapiro AMJ, et al. Xiap overexpression in human islets prevents early posttransplant apoptosis and reduces the islet mass needed to treat diabetes. Diabetes. 2005 2005/09//:2541+. [CrossRef]
- Emamaullee JA, Stanton L, Schur C, Shapiro AMJ. Caspase inhibitor therapy enhances marginal mass islet graft survival and preserves long-term function in islet transplantation. Diabetes. 2007 2007/05//:1289+. [CrossRef]
- Emamaullee JA, Davis J, Pawlick R, Toso C, Merani S, Cai S-X, et al. The caspase selective inhibitor ep1013 augments human islet graft function and longevity in marginal mass islet transplantation in mice. Diabetes. 2008; 54: 2541-2548. [CrossRef]
- Brandhorst D, Kumarasamy V, Maatoui A, Alt A, Bretzel RG, Brandhorst H. Porcine islet graft function is affected by pretreatment with a caspase-3 inhibitor. Cell Transplant. 2006; 15: 311-317. [CrossRef]
- Aikin R, Rosenberg L, Paraskevas S, Maysinger D. Inhibition of caspase-mediated parp-1 cleavage results in increased necrosis in isolated islets of langerhans. J Mol Med (Berl). 2004; 82: 389-397. [CrossRef]
- Brandhorst D, Brandhorst H, Maataoui V, Maataoui A, Johnson PR. Anti-caspase-3 preconditioning increases proinsulin secretion and deteriorates posttransplant function of isolated human islets. Apoptosis. 2013; 18: 681-688. [CrossRef]
- Baker MS, Chen X, Rotramel AR, Nelson JJ, Lu B, Gerard C, et al. Genetic deletion of chemokine receptor cxcr3 or antibody blockade of its ligand ip-10 modulates posttransplantation graft-site lymphocytic infiltrates and prolongs functional graft survival in pancreatic islet allograft recipients. Surgery. 2003; 134: 126-133. [CrossRef]
- Posselt AM, Bellin MD, Tavakol M, Szot GL, Frassetto LA, Masharani U, et al. Islet transplantation in type 1 diabetics using an immunosuppressive protocol based on the antilfa-1 antibody efalizumab. Am J Transplant. 2010; 10: 1870-1880. [CrossRef]
- McCall M, Pawlick R, Kin T, Shapiro AMJ. Anakinra potentiates the protective effects of etanercept in transplantation of marginal mass human islets in immunodeficient mice. Am J Transplant. 2011; 12: 322-329. [CrossRef]
- Gmyr V, Bonner C, Moerman E, Tournoys A, Delalleau N, Quenon A, et al. Human recombinant antithrombin (atryn®) administration improves survival and prevents intravascular coagulation after intraportal islet transplantation in a piglet model. Cell Transplant. 2017; 26: 309-317. [CrossRef]
- Mark L. Kahn MN-M, Michael J. Shapiro, Hiroaki Ishihara, and Shaun R. Coughlin. Proteaseactivated receptors 1 and 4 mediate activation of human platelets by thrombin. J Clin Invest. 1999; 103: 879-887. [CrossRef]
- Coughlin SR. Thrombin signalling and protease-activated receptors. Nature. 2000; 407: 258. [CrossRef]
- Jack Hirsh VF. Guide to anticoagulant therapy. Part 1: Heparin. Circulation. 1994; 89: 14491468. [CrossRef]
- Markwardt F. The development of hirudin as an antithrombotic drug. Thromb Res. 1994; 74: 1-23. [CrossRef]
- Elg M, Gustafsson D, Carlsson S. Antithrombotic effects and bleeding time of thrombin inhibitors and warfarin in the rat. Thromb Res. 1999; 94: 187-197. [CrossRef]
- Eriksson H, Eriksson UG, Frison L, Hansson PO, Held P, Holmström M, et al. Pharmacokinetics and pharmacodynamics of melagatran, a novel synthetic lmw thrombin inhibitor, in patients with acute dvt. Thromb Haemost. 1999; 81: 358-363. [CrossRef]
- Keisu M, Andersson TB. Drug-induced liver injury in humans: The case of ximelagatran. Handb Exp Pharmacol. 2010: 407-418. [CrossRef]
- Matsuda S, Koyasu S. Mechanisms of action of cyclosporine. Immunopharmacology. 2000; 47: 119-125. [CrossRef]
- Nakajima Y, Kessler M, Nakano H, Lie TS. Restoration of t cell responsiveness to interleukin-2 in recipients of pancreatic islet xenografts treated with cyclosporine. Transplantation. 1985; 40: 73-76. [CrossRef]
- Plosker GL, Foster RH. Tacrolimus. Drugs. 2000; 59: 323-389. [CrossRef]
- Hirano Y, Hirano Y, Fujihira S, Fujihira S, Ohara K, Ohara K, et al. Morphological and functional changes of islets of langerhans in fk506- treated rats. Transplantation. 1992; 53: 889-894. [CrossRef]
- Fabian MC, Fabian MC, Lakey JRT, Lakey JRT, Rajotte RV, Rajotte RV, et al. The efficacy and toxicity of rapamycin in murine islet transplantation: In vitro and in vivo studies. Transplantation. 1993; 56: 1137-1142. [CrossRef]
- Ransom JT, Ransom JT. Mechanism of action of mycophenolate mofetil. Ther Drug Monit. 1995; 17: 681-684. [CrossRef]
- Wennberg L, Karlsson-Parra A, Sundberg B, Rafael E, Liu J, Zhu S, et al. Efficacy of immunosuppressive drugs in islet xenotransplantation: Leflunomide in combination with cyclosporine and mycophenolate mofetil prevents islet xenograft rejection in the pig-to-rat model. Transplantation. 1997; 63: 1234-1242. [CrossRef]
- Waldmann TA, O'Shea J. The use of antibodies against the il-2 receptor in transplantation. Curr Opin Immunol. 1998; 10: 507-512. [CrossRef]
- Brandhorst D, Brandhorst H, Zwolinski A, Nahidi F, Bretzel RG. Prevention of early islet graft failure by selective inducible nitric oxide synthase inhibitors after pig to nude rat intraportal islet transplantation. Transplantation. 2001; 71: 179-184. [CrossRef]
- Montolio M, Téllez N, Biarnés M, Soler J, Montanya E. Short-term culture with the caspase inhibitor z-vad.Fmk reduces beta cell apoptosis in transplanted islets and improves the metabolic outcome of the graft. Cell Transplant. 2005; 14: 59-65. [CrossRef]
- Yang W, Guastella J, Huang J-C, Wang Y, Zhang L, Xue D, et al. Mx1013, a dipeptide caspase inhibitor with potent in vivo antiapoptotic activity. Br J Pharmacol. 2003; 140: 402-412. [CrossRef]
- Goffe B, Cather JC. Etanercept: An overview. J Am Acad Dermatol. 2003; 49: 105-111. [CrossRef]
- Fleischmann R, Stern R, Iqbal I. Anakinra: An inhibitor of il-1 for the treatment of rheumatoid arthritis. Expert Opin Biol Ther. 2004; 4: 1333-1344. [CrossRef]
- Schwarznau A, Hanson MS, Sperger JM, Schram BR, Danobeitia JS, Greenwood KK, et al. Il-1β receptor blockade protects islets against pro-inflammatory cytokine induced necrosis and apoptosis. J Cell Physiol. 2009; 220: 341-347. [CrossRef]
- Mohan R, Hammers H, Bargagna-mohan P, Zhan X, Herbstritt C, Ruiz A, et al. Withaferin a is a potent inhibitor of angiogenesis. Angiogenesis. 2004; 7: 115-122. [CrossRef]
- Peng H, Olsen G, Tamura Y, Noguchi H, Matsumoto S, Levy MF, et al. Inhibition of inflammatory cytokine-induced response in human islet cells by withaferin a. Transplant Proc. 2010; 42: 2058-2061. [CrossRef]
- Chang CA, Kanak MA, Yoshimatsu G, Lawrence MC, Kane RR, Naziruddin B, editors. A small molecule inhibitor of toll-like receptor-4 (tlr-4) effectively protects islets from ibmir2015; PHILADELPHIA: LIPPINCOTT WILLIAMS & WILKINS.
- Vaithilingam V, Tuch BE. Islet transplantation and encapsulation: An update on recent developments. Rev Diabet Stud. 2011; 8: 51-67. [CrossRef]
- Heintz RE, Merideth N, Murphy M, Moloney MK, et al. Insulin independence in a type 1 diabetic patient after encapsulated islet transplantation. Lancet. 1994; 343: 950-951. [CrossRef]
- Scharp DW, Marchetti P. Encapsulated islets for diabetes therapy: History, current progress, and critical issues requiring solution. Adv Drug Deliv Rev. 2014; 67-68: 35-73. [CrossRef]
- Tuch BE, Keogh GW, Williams LJ, Wu W, Foster JL, Vaithilingam V, et al. Safety and viability of microencapsulated human islets transplanted into diabetic humans. Diabetes Care. 2009; 32: 1887-1889. [CrossRef]
- Hillberg Anna L, Oudshoorn M, Lam Janice BB, Kathirgamanathan K. Encapsulation of porcine pancreatic islets within an immunoprotective capsule comprising methacrylated glycol chitosan and alginate. J Biomed Mater Res B Appl Biomater. 2014; 103: 503-518. [CrossRef]
- Sawhney AS, Pathak CP, Hubbell JA. Interfacial photopolymerization of poly(ethylene glycol)based hydrogels upon alginate-poly(l-lysine) microcapsules for enhanced biocompatibility. Biomaterials. 1993; 14: 1008-1016. [CrossRef]
- Sefton MV, Stevenson WTK. Microencapsulation of live animal-cells using polyacrylates. Adv Polym Sci. 1993; 107: 143-197. [CrossRef]
- Peterson KP, Peterson CM, Pope EJA. Silica sol-gel encapsulation of pancreatic islets. Proc Soc Exp Biol Med. 1998; 218: 365-369. [CrossRef]
- Lee BR, Hwang JW, Choi YY, Wong SF, Hwang YH, Lee DY, et al. In situ formation and collagenalginate composite encapsulation of pancreatic islet spheroids. Biomaterials. 2012; 33: 837845. [CrossRef]
- Sobol M, Bartkowiak A, de Haan B, de Vos P. Cytotoxicity study of novel water-soluble chitosan derivatives applied as membrane material of alginate microcapsules. J Biomed Mater Res A. 2012; 101A: 1907-1914. [CrossRef]
- Steele Joseph AM, Barron Annelise E, Carmona E, Hallé J-P, Neufeld Ronald J. Encapsulation of protein microfiber networks supporting pancreatic islets. J Biomed Mater Res A. 2012; 100A: 3384-3391. [CrossRef]
- Dumpala PR, Holdcraft RW, Martis PC, Laramore MA, Parker TS, Levine DM, et al. Retention of gene expression in porcine islets after agarose encapsulation and long-term culture. Biochem Biophys Res Commun. 2016; 476: 580-585. [CrossRef]
- Wang R, Rosenberg L. Maintenance of beta-cell function and survival following islet isolation requires re-establishment of the islet-matrix relationship. J Endocrinol. 1999; 163: 181-190. [CrossRef]
- Algire GH, Algire GH, Weaver JM, Weaver JM, Prehn RT, Prehn RT. Growth of cells in vivo in diffusion chambers. I. Survival of homografts in immunized mice. J Natl Cancer Inst. 1954; 15: 493-507.
- Prehn RT, Prehn RT, Weaver JM, Weaver JM, Algire GH, Algire GH. The diffusion-chamber technique applied to a study of the nature of homograft resistance. J Natl Cancer Inst. 1954; 15: 509-517.
- Gates RJ, Gates R, Smith R, Hunt MI, Smith R, Hunt MI, et al. Return to normal of bloodglucose, plasma-insulin, and weight gain in new zealand obese mice after implantation of islets of langerhans. Lancet. 1972; 2;300;: 567-570. [CrossRef]
- Strautz RL. Studies of hereditary-obese mice (obob) after implantation of pancreatic islets in millipore filter capsules. Diabetologia. 1970; 6: 306-312. [CrossRef]
- Brauker JH, Carr-Brendel VE, Martinson LA, Crudele J, Johnston WD, Johnson RC. Neovascularization of synthetic membranes directed by membrane microarchitecture. J Biomed Mater Res. 1995; 29: 1517. [CrossRef]
- Nyitray CE, Chang R, Faleo G, Lance KD, Bernards DA, Tang Q, et al. Polycaprolactone thin-film micro- and nanoporous cell-encapsulation devices. ACS Nano. 2015; 9: 5675-5682. [CrossRef]
- Mendelsohn A, Desai T, editors. Inorganic nanoporous membranes for immunoisolated cellbased drug delivery. Adv Exp Med Biol. 2010; 670: 104-125. [CrossRef]
- Dang TT, Thai AV, Cohen J, Slosberg JE, Siniakowicz K, Doloff JC, et al. Enhanced function of immuno-isolated islets in diabetes therapy by co-encapsulation with an anti-inflammatory drug. Biomaterials. 2013; 34: 5792-5801. [CrossRef]
- Allen J, Ryu J, Maggi A, Flores B, Greer JR, Desai T. Tunable microfibers suppress fibrotic encapsulation via inhibition of tgf[beta] signaling. Tissue Eng Part A. 2016; 22: 142-150. [CrossRef]
- Lim F, Sun AM. Microencapsulated islets as bioartificial endocrine pancreas. Science. 1980; 210: 908-910. [CrossRef]
- O'Shea GM, Goosen MF, Sun AM. Prolonged survival of transplanted islets of langerhans encapsulated in a biocompatible membrane. Biochim Biophys Acta. 1984; 804: 133. [CrossRef]
- Klöck G, Frank H, Houben R, Zekorn T, Horcher A, Siebers U, et al. Production of purified alginates suitable for use in immunoisolated transplantation. Appl Microbiol Biotechnol. 1994; 40: 638. [CrossRef]
- Otterlei M, Ostgaard K, Skjåk-Braek G, Smidsrød O, Soon-Shiong P, Espevik T. Induction of cytokine production from human monocytes stimulated with alginate. J Immunother. 1991; 10: 286. [CrossRef]
- Rokstad AM, Brekke O-L, Steinkjer B, Ryan L, Kolláriková G, Strand BL, et al. Alginate microbeads are complement compatible, in contrast to polycation containing microcapsules, as revealed in a human whole blood model. Acta Biomater. 2011; 7: 2566-2578. [CrossRef]
- Wang T, Lacík I, Brissová M, Anilkumar AV, Prokop A, Hunkeler D, et al. An encapsulation system for the immunoisolation of pancreatic islets. Nat Biotechnol. 1997; 15: 358. [CrossRef]
- Foster GA, García AJ. Bio-synthetic materials for immunomodulation of islet transplants. Adv Drug Deliv Rev. 2017; 114: 266-271. [CrossRef]
- Chen J-P, Chu IM, Shiao M-Y, Hsu BR-S, Fu S-H. Microencapsulation of islets in peg-amine modified alginate-poly(l-lysine)-alginate microcapsules for constructing bioartificial pancreas. J Ferment Bioeng. 1998; 86: 185-190. [CrossRef]
- Weber LM, Lopez CG, Anseth KS. Effects of peg hydrogel crosslinking density on protein diffusion and encapsulated islet survival and function. J Biomed Mater Res A. 2008; 90A: 720729. [CrossRef]
- Weber LM, Anseth KS. Hydrogel encapsulation environments functionalized with extracellular matrix interactions increase islet insulin secretion. Matrix Biol. 2008; 27: 667-673. [CrossRef]
- Tomei AA, Manzoli V, Fraker CA, Giraldo J, Velluto D, Najjar M, et al. Device design and materials optimization of conformal coating for islets of langerhans. Proc Natl Acad Sci USA. 2014; 111: 10514-10519. [CrossRef]
- Evron Y, Colton CK, Ludwig B, Weir GC, Zimermann B, Maimon S, et al. Long-term viability and function of transplanted islets macroencapsulated at high density are achieved by enhanced oxygen supply. Sci Rep. 2018; 8: 1-13. [CrossRef]
- Papas KK, Avgoustiniatos ES, Suszynski TM. Effect of oxygen supply on the size of implantable islet-containing encapsulation devices. Panminerva Med. 2016; 58: 72-77.
- Suszynski TM, Avgoustiniatos ES, Papas KK. Intraportal islet oxygenation. J Diabetes Sci Technol. 2014; 8: 575-580. [CrossRef]
- Zisch AH, Lutolf MP, Ehrbar M, Raeber GP, Rizzi SC, Davies N, et al. Cell-demanded release of vegf from synthetic, biointeractive cell ingrowth matrices for vascularized tissue growth. FASEB J. 2003; 17: 2260-2262. [CrossRef]
- Phelps EA, Templeman KL, Thulé PM, García AJ. Engineered vegf-releasing peg–mal hydrogel for pancreatic islet vascularization. Drug Deliv Transl Res. 2015; 5: 125-136. [CrossRef]
- Bloch K, Papismedov E, Yavriyants K, Vorobeychik M, Beer S, Vardi P. Photosynthetic oxygen generator for bioartificial pancreas. Tissue Eng. 2006; 12: 337-344. [CrossRef]
- Ludwig B, Reichel A, Steffen A, Zimerman B, Schally AV, Block NL, et al. Transplantation of human islets without immunosuppression. Proc Natl Acad Sci USA. 2013; 110: 19054-19058. [CrossRef]
- Brauker J, Martinson LA, Young SK, Johnson RC. Local inflammatory response around diffusion chambers containing xenografts. Nonspecific destruction of tissues and decreased local vascularization. Transplantation. 1996; 61: 1671. [CrossRef]
- Pedraza E, Coronel MM, Fraker CA, Ricordi C, Stabler CL. Preventing hypoxia-induced cell death in beta cells and islets via hydrolytically activated, oxygen-generating biomaterials. Proc Natl Acad Sci USA. 2012; 109: 4245-4250. [CrossRef]
- Kumagai-Braesch M, Jacobson S, Mori H, Jia X, Takahashi T, Wernerson A, et al. The theracyte™ device protects against islet allograft rejection in immunized hosts. Cell Transplant. 2013; 22: 1137-1146. [CrossRef]
- Krieger JR, Ogle ME, McFaline-Figueroa J, Segar CE, Temenoff JS, Botchwey EA. Spatially localized recruitment of anti-inflammatory monocytes by sdf-1α-releasing hydrogels enhances microvascular network remodeling. Biomaterials. 2016; 77: 280-290. [CrossRef]
- Weaver JD, Headen DM, Hunckler MD, Coronel MM, Stabler CL, García AJ. Design of a vascularized synthetic poly(ethylene glycol) macroencapsulation device for islet transplantation. Biomaterials. 2018; 172: 54-65. [CrossRef]
- Liu JMH, Zhang J, Zhang X, Hlavaty KA, Ricci CF, Leonard JN, et al. Transforming growth factorbeta 1 delivery from microporous scaffolds decreases inflammation post-implant and enhances function of transplanted islets. Biomaterials. 2016; 80: 11-19. [CrossRef]
- Karin N. The multiple faces of cxcl12 (sdf-1α) in the regulation of immunity during health and disease. J Leukoc Biol. 2010; 88: 463-473. [CrossRef]
- Sánchez-Martín L, Estecha A, Samaniego R, Sánchez-Ramón S, Vega MÁ, Sánchez-Mateos P. The chemokine cxcl12 regulates monocyte-macrophage differentiation and runx3 expression. Blood. 2011; 117: 88-97. [CrossRef]
- Chen T, Yuan J, Duncanson S, Hibert ML, Kodish BC, Mylavaganam G, et al. Alginate encapsulant incorporating cxcl12 supports long-term allo- and xenoislet transplantation without systemic immune suppression. Am J Transplant. 2015; 15: 618-627. [CrossRef]
- Xu X, Zhu F, Zhang M, Zeng D, Luo D, Liu G, et al. Stromal cell-derived factor-1 enhances wound healing through recruiting bone marrow-derived mesenchymal stem cells to the wound area and promoting neovascularization. Cells Tissues Organs. 2013; 197: 103-113. [CrossRef]
- Liu Z, Habener JF. Stromal cell-derived factor-1 promotes survival of pancreatic beta cells by the stabilisation of beta-catenin and activation of transcription factor 7-like 2 (tcf7l2). Diabetologia. 2009; 52: 1589-1598. [CrossRef]
- Su J, Hu B-H, Lowe WL, Kaufman DB, Messersmith PB. Anti-inflammatory peptidefunctionalized hydrogels for insulin-secreting cell encapsulation. Biomaterials. 2010; 31: 308314. [CrossRef]
- Robertson RP, Harmon JS. Pancreatic islet β-cell and oxidative stress: The importance of glutathione peroxidase. FEBS Lett. 2007; 581: 3743-3748. [CrossRef]
- Pi J, Bai Y, Zhang Q, Wong V, Floering LM, Daniel K, et al. Reactive oxygen species as a signal in glucose-stimulated insulin secretion. Diabetes. 2007 2007/07//:1783+. [CrossRef]
- Barkai U, Weir GC, Colton CK, Ludwig B, Bornstein SR, Brendel MD, et al. Enhanced oxygen supply improves islet viability in a new bioartificial pancreas. Cell Transplant. 2013; 22: 14631476. [CrossRef]
- Carlsson P-O, Espes D, Sedigh A, Rotem A, Zimerman B, Grinberg H, et al. Transplantation of macroencapsulated human islets within the bioartificial pancreas βair to patients with type 1 diabetes mellitus. Am J Transplant. 2017; 18: 1735-1744. [CrossRef]
- Baidal DA, Ricordi C, Berman DM, Alvarez A, Padilla N, Ciancio G, et al. Bioengineering of an intraabdominal endocrine pancreas. N Engl J Med. 2017; 376: 1887-1889. [CrossRef]
- Gala-Lopez BL, Dinyari P, Malcolm AJ, Kin T, Pawlick LR, Senior PA, Shapiro AMJ. Subcutaneous clinical islet transplantation in a prevascularized subcutaneous pouchpreliminary experience. CellR4. 2016; 4: e2132.
- Morozov VA, Wynyard S, Matsumoto S, Abalovich A, Denner J, Elliott R. No perv transmission during a clinical trial of pig islet cell transplantation. Virus Res. 2017; 227: 34-40. [CrossRef]
- Schulz TC. Concise review: Manufacturing of pancreatic endoderm cells for clinical trials in type 1 diabetes. Stem Cells Transl Med. 2015; 4: 927-931. [CrossRef]
- Berman DM, Molano RD, Fotino C, Ulissi U, Gimeno J, Mendez AJ, et al. Bioengineering the endocrine pancreas: Intraomental islet transplantation within a biologic resorbable scaffold. Diabetes. 2016; 65: 1350-1361. [CrossRef]
- Panza JL, R. Wagner W, Rilo HLR, Harsha Rao R, Beckman EJ, Russell AJ. Treatment of rat pancreatic islets with reactive peg. Biomaterials. 2000; 21: 1155-1164. [CrossRef]
- Dong Yun L, Sang Jin P, Nam JH, Byun Y. A new strategy toward improving immunoprotection in cell therapy for diabetes mellitus: Long-functioning pegylated islets in vivo. Tissue Eng. 2006; 12: 615-623. [CrossRef]
- Hashemi J, Hashemi-Najafabadi S, Vasheghani-Farahani E. Synergistic effect of pegylation and pentoxifylline addition on immunoprotection of pancreatic islets. J Biomater Sci Polym Ed. 2017; 28: 33-49. [CrossRef]
- Jang JY, Lee DY, Park SJ, Byun Y. Immune reactions of lymphocytes and macrophages against peg-grafted pancreatic islets. Biomaterials. 2004; 25: 3663-3669. [CrossRef]
- Yun Lee D, Hee Nam J, Byun Y. Functional and histological evaluation of transplanted pancreatic islets immunoprotected by pegylation and cyclosporine for 1 year. Biomaterials. 2007; 28: 1957-1966. [CrossRef]
- Teramura Y, Iwata H. Improvement of graft survival by surface modification with poly(ethylene glycol)-lipid and urokinase in intraportal islet transplantation. Transplantation. 2011; 91: 271-278. [CrossRef]
- Teramura Y, Iwata H. Surface modification of islets with peg-lipid for improvement of graft survival in intraportal transplantation. Transplantation. 2009; 88: 624-630. [CrossRef]
- Xie D, Smyth CA, Eckstein C, Bilbao G, Mays J, Eckhoff DE, et al. Cytoprotection of pegmodified adult porcine pancreatic islets for improved xenotransplantation. Biomaterials. 2005; 26: 403-412. [CrossRef]
- Scott MD, Chen AM. Beyond the red cell: Pegylation of other blood cells and tissues. Transfus Clin Biol. 2004; 11: 40-46. [CrossRef]
- Stabler CL, Sun X-L, Cui W, Wilson JT, Haller CA, Chaikof EL. Surface re-engineering of pancreatic islets with recombinant azido-thrombomodulin. Bioconjug Chem. 2007; 18: 17131715. [CrossRef]
- Wilson JT, Haller CA, Qu Z, Cui W, Urlam MK, Chaikof EL. Biomolecular surface engineering of pancreatic islets with thrombomodulin. Acta Biomater. 2010; 6: 1895-1903. [CrossRef]
- Cabric S, Sanchez J, Lundgren T, Foss A, Felldin M, Källen R, et al. Islet surface heparinization prevents the instant blood-mediated inflammatory reaction in islet transplantation. Diabetes. 2007; 56: 2008-2015. [CrossRef]
- Im B-H, Jeong J-H, Haque MR, Lee DY, Ahn C-H, Kim JE, et al. The effects of 8-arm-pegcatechol/heparin shielding system and immunosuppressive drug, fk506 on the survival of intraportally allotransplanted islets. Biomaterials. 2013; 34: 2098-2106. [CrossRef]
- Chang CA, Akinbobuyi B, Quintana JM, Yoshimatsu G, Naziruddin B, Kane RR. Ex-vivo generation of drug-eluting islets improves transplant outcomes by inhibiting tlr4-mediated nfkb upregulation. Biomaterials. 2018; 159: 13-24. [CrossRef]
- Jeong J-H, Hong SW, Hong S, Yook S, Jung Y, Park J-B, et al. Surface camouflage of pancreatic islets using 6-arm-peg-catechol in combined therapy with tacrolimus and anti-cd154 monoclonal antibody for xenotransplantation. Biomaterials. 2011; 32: 7961-7970. [CrossRef]
- Giraldo JA, Molano RD, Rengifo HR, Fotino C, Gattás-Asfura KM, Pileggi A, et al. The impact of cell surface pegylation and short-course immunotherapy on islet graft survival in an allogeneic murine model. Acta Biomater. 2017; 49: 272-283. [CrossRef]
- Teramura Y, Kaneda Y, Iwata H. Islet-encapsulation in ultra-thin layer-by-layer membranes of poly(vinyl alcohol) anchored to poly(ethylene glycol)–lipids in the cell membrane. Biomaterials. 2007; 28: 4818-4825. [CrossRef]
- SoRelle JA, Kanak MA, Itoh T, Horton JM, Naziruddin B, Kane RR. Comparison of surface modification chemistries in mouse, porcine, and human islets. J Biomed Mater Res A. 2015; 103: 869-877. [CrossRef]
- Santi DV, Schneider EL, Reid R, Robinson L, Ashley GW. Predictable and tunable half-life extension of therapeutic agents by controlled chemical release from macromolecular conjugates. Proc Natl Acad Sci USA. 2012; 109: 6211-6216. [CrossRef]
- Jung YS, Jeong J-H, Yook S, Im B-H, Seo J, Hong SW, et al. Surface modification of pancreatic islets using heparin-dopa conjugate and anti-cd154 mab for the prolonged survival of intrahepatic transplanted islets in a xenograft model. Biomaterials. 2012; 33: 295-303. [CrossRef]
- Kristensen EME, Rensmo H, Larsson R, Siegbahn H. Characterization of heparin surfaces using photoelectron spectroscopy and quartz crystal microbalance. Biomaterials. 2003; 24: 41534159. [CrossRef]
- Hirsh J, Warkentin TE, Shaughnessy SG, Anand SS, Halperin JL, Raschke R, et al. Heparin and low-molecular-weight heparin mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety. CHEST. 2001; 119: 64S-94S. [CrossRef]
- Shimoda M, Chen S, Noguchi H, Matsumoto S, Grayburn PA. In vivo non-viral gene delivery of human vasular endothelial growth factor imporves human islet survival and function after transplantation. Xenotransplantation. 2009; 16: 1.
- Schlessinger J, Plotnikov AN, Ibrahimi OA, Eliseenkova AV, Yeh BK, Yayon A, et al. Crystal structure of a ternary fgf-fgfr-heparin complex reveals a dual role for heparin in fgfr binding and dimerization. Mol Cell. 2000; 6: 743-750. [CrossRef]
- Yolcu ES, Zhao H, Bandura-Morgan L, Lacelle C, Woodward KB, Askenasy N, et al. Pancreatic islets engineered with sa-fasl protein establish robust localized tolerance by inducing regulatory t cells in mice. J Immunol. 2011; 187: 5901. [CrossRef]
- Izadi Z, Hajizadeh-Saffar E, Hadjati J, Habibi-Anbouhi M, Ghanian MH, Sadeghi-Abandansari H, et al. Tolerance induction by surface immobilization of jagged-1 for immunoprotection of pancreatic islets. Biomaterials. 2018; 182: 191-201. [CrossRef]
- Yvon ES, Vigouroux S, Rousseau RF, Biagi E, Amrolia P, Dotti G, et al. Overexpression of the notch ligand, jagged-1, induces alloantigen-specific human regulatory t cells. Blood. 2003; 102: 3815. [CrossRef]
- Hwang YH, Kim MJ, Lee DY. Mri-sensitive contrast agent with anticoagulant activity for surface camouflage of transplanted pancreatic islets. Biomaterials. 2017; 138: 121-130. [CrossRef]
- Shapiro AM, Pokrywczynska M, Ricordi C. Clinical pancreatic islet transplantation. Nat Rev Endocrinol. 2017; 13: 268-277. [CrossRef]
- Gamble A, Pepper AR, Bruni A, Shapiro AMJ. The journey of islet cell transplantation and future development. Islets. 2018; 10: 80-94. [CrossRef]