Changes in HLA-G Expression by Leucocyte Subsets Following Renal Transplantation and the Effect of Immunosuppressive Drugs on Leucocyte HLA-G Expression in Vitro
Zaid Albayati 1 ![]() , Sally Heyworth 2
, Sally Heyworth 2 ![]() , Jane Moberly 2
, Jane Moberly 2 ![]() , Derek Middleton 3
, Derek Middleton 3 ![]() , Brian Flanagan 4
, Brian Flanagan 4 ![]() , Abdul Hammad 2
, Abdul Hammad 2 ![]() , Stephen E Christmas 1, *
, Stephen E Christmas 1, * ![]()
- Department of Clinical Infection, Microbiology & Immunology, Institute of Infection & Global Health, University of Liverpool, UK
- Transplant Unit, Royal Liverpool and Broadgreen Hospital NHS Trust, Liverpool, UK
- Transplant Immunology, Royal Liverpool and Broadgreen Hospital NHS Trust, Liverpool, UK
- Women’s and Children’s Health, Institute of Translational Medicine, University of Liverpool, UK
* Correspondence: Stephen E Christmas![]()
Academic Editor: Yasuhiko Sugawara
Received: October 10, 2018 | Accepted: December 12, 2018 | Published: December 14, 2018
OBM Transplantation 2018, Volume 2, Issue 4 doi:10.21926/obm.transplant.1804032
Recommended citation: Albayati Z, Heyworth S, Moberly J, Middleton D, Flanagan B, Hammad A, Christmas SF. Changes in HLA-G Expression by Leucocyte Subsets Following Renal Transplantation and the Effect of Immunosuppressive Drugs on Leucocyte HLA-G Expression in Vitro. OBM Transplantation 2018; 2(4): 032; doi:10.21926/obm.transplant.1804032.
© 2018 by the authors. This is an open access article distributed under the conditions of the Creative Commons by Attribution License, which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is correctly cited.
Abstract
Background: HLA-G is an immunosuppressive molecule with a potential role in allograft acceptance.
Methods: Expression of HLA-G on leucocyte subpopulations was studied in a group of 21 renal transplant patients immediately prior to and 2 months post-transplantation.
Results: Significant increases in proportions of total HLA-G+CD4+ T cells were seen (0.5+/-0.1% to 7.9+/-3.2%; p<0.01) and also for CD45RA+, CD45RO+ and CD69+ subsets. Increases in proportions of HLA-G+ cells were also seen in CD8+ T cells and their subsets, CD56+ T cells, B cells, NK cells and monocytes, and also of serum soluble HLA-G, but these did not reach statistical significance. When leucocytes from healthy subjects were cultured for 7 days in therapeutic concentrations of tacrolimus and mycophenolate mofetil, no significant increases in proportions of HLA-G+CD4 or CD8+ T cells were found while those for CD56+ T cells (3.4+/-0.2% to 13.0+/-0.4%) and B cells (1.6+/-0.1% to 6.4+/-0.2%) increased significantly (p<0.01). Proportions of HLA-G+CD103+ B cells, NK cells and CD56+ T cells, but not classical T cells, were also significantly increased (p<0.05) following culture in immunosuppressive drugs.
Conclusions: The results show that HLA-G is upregulated in leucocyte subsets following renal transplantation but that in CD4+ and CD8+ T cells this is not directly mediated by immunosuppressive drugs.
Keywords
HLA-G; T lymphocytes; B lymphocytes; NK cells; KIR2DL4; tacrolimus; kidney transplantation
1. Introduction
HLA-G is a class 1b HLA molecule normally expressed at high levels on extravillous cytotrophoblast at the materno-fetal interface [1]. Here, its function is thought to be concerned with preventing rejection of the fetus by the maternal immune system [2], either by direct inhibition of NK cell and/or T cell function, by trogocytosis [3] or secretion of soluble HLA-G and uptake by other cell types [4]. Ligands for HLA-G include ILT2/LILRB1 and ILT4/LILRB2, mainly expressed on lymphocyte and monocyte subsets, and KIR2DL4, predominantly expressed on NK cells [5].
Expression of HLA-G has previously been studied in relation to clinical cardiac transplantation where endomyocardial expression [6,7] and levels of soluble HLA-G were associated with graft acceptance [7,8]. Serum levels increased post-transplantation in patients with good graft function but were significantly decreased in those suffering from cardiac allograft vasculopathy [9]. However, levels of soluble HLA-G were also found to be increased in patients suffering from heart failure [10]. In lung transplant patients, bronchial epithelial expression of HLA-G was more prominent in patients with good graft function than in those with signs of acute rejection and anti-HLA antibodies [11,12]. Similarly, in liver transplant patients increased levels of sHLA-G were associated with favourable graft function [13].
In renal transplant patients, peripheral blood mononuclear cell HLA-G expression at the mRNA level was higher in patients with stable renal function than in those with acute rejection [14,15]. However, HLA-G mRNA levels in renal biopsies and protein expression on tubular epithelium were higher in patients with acute rejection than those with non-rejected but dysfunctional grafts [16]. At the protein level, HLA-G expression by tubular epithelium was higher in patients without rejection [17]. At the genomic level, dimorphisms 3’ to the HLA-G gene, and associated with increased degradation of HLA-G mRNA, and hence lower levels of HLA-G expression, were proportionately increased in kidney transplant patients with rejection [18,19,20,21]. However, this was not the case in all studies [22,23] and the opposite effect was reported for liver transplant patients [24]. Interestingly, kidney transplant patients with complete HLA-G matches were less likely to experience rejection episodes [25], although this could be related to linked HLA alleles rather than to HLA-G itself.
In several studies, serum soluble HLA-G levels were reported to be increased in kidney transplant patients with good renal function compared to those with rejection [26,27,28,29,30,31,32]. This may be related to the presence of soluble or monocyte-bound HLA-G dimers [33]. However, patients with acute tubular necrosis were also reported to have elevated serum sHLA-G levels [34].
The observed increases in HLA-G expression in organ transplant patients may be related to the ongoing allogeneic response and studies in vitro have shown that HLA-G is upregulated on allostimulated CD4+ and CD8+ T cells as well as in transplant patients in vivo [35]. Such changes in HLA-G expression may potentially also be related to the immunosuppressive drug treatment or other factors, such as activation of cytomegalovirus (CMV). In previous work in healthy subjects we have shown that stimulation of peripheral blood mononuclear cells with CMV antigens resulted in increased proportions of leucocytes expressing HLA-G, particularly B cells and CD56+ T cells [36]. The aims of the present experiments were to assess any post-transplant changes in HLA-G expression by a range of leucocyte subsets in renal transplant patients. The role of standard immunosuppressive medications in mediating any changes observed was also studied in vitro in leucocytes from healthy subjects. The hypothesis was that HLA-G expression would be increased in patients with surviving transplants and that immunosuppressive drugs may mediate any increases.
2. Materials and Methods
2.1 Ethics Statement
Renal transplant patients were recruited from the Transplant Unit, Royal Liverpool and Broadgreen University Hospital Trust between 2015 and 2017. Patients gave informed consent for blood donations and ethical approval for the study was obtained from Newcastle and North Tyneside NREC (14/NE/0094) and Haydock NRES Committee North West (15/NW/0351). Healthy subjects were recruited from the department and ethical approval was obtained from the University of Liverpool Interventional Ethics Committee (RETH000685).
2.2 Transplant Patients and Samples
Patients with renal failure were transplanted with a kidney from a deceased or living related donor using standard protocols. Maintenance therapy was a combination of Prograf® and mycophenolate mofetil. Patients at risk of cytomegalovirus (CMV) re-activation were given prophylactic ganciclovir treatment for 3 months post-transplantation. None of the patients studied experienced a rejection episode or an episode of CMV or BK virus re-activation during the course of the study period.
2.3 Blood Sampling
Blood samples were taken immediately pre-transplantation and at 3 and 6 months post-transplantation into EDTA, mononuclear leucocytes purified by ficoll density gradient centrifugation and plasma was stored at -20ºC until use.
2.4 Mononuclear Cell Preparation and Flow Cytometry
Mononuclear cells accumulating at the plasma-ficoll interface were washed and resuspended in RPMI-1640 + 10% heat-inactivated fetal calf serum + 2mM L-glutamine antibiotics (complete medium; CM) at 106 cells/ml. For cell surface phenotypic analysis 4 x 105 cells in a volume of 100µl were aliquoted into individual tubes and combinations of 4µl of fluorescently conjugated monoclonal antibodies (mAb) added to a final concentration of 10µg/ml. The panel of mAb used is shown in Table 1, together with isotype controls. Cells were labelled for 30min at 4ºC, washed and resuspended in 1ml ice cold phosphate buffered isotonic saline (PBS) prior to analysis. Cells were then run through an Accuri C6 flow cytometer (Becton Dickinson, Oxford, UK), gating for lymphocytes or monocytes based upon forward and side scatter parameters. 5 x 104 cells were acquired per sample and an appropriate isotype control was run. The fluorescence threshold was set at a point at which <1% of cells from the isotype control were positive and this was subtracted from the value obtained with labelled cells. Up to four labelled mAb conjugated to appropriate fluorochromes were used for each sample, with appropriate colour compensation. Results were expressed as % positive cells in relation to total lymphocytes or monocytes, or, for 2- or 3-colour staining as a percentage of the relevant lymphocyte subpopulation.
Table 1 Monoclonal antibodies (mAb) used in this study
|
Monoclonal antibody |
Isotype |
Code # |
Supplier |
|
Anti-CD3-FITC |
IgG2a |
317306 |
Biolegend, London, UK |
|
Anti-CD4-PE |
IgG2b |
317410 |
Biolegend, London, UK |
|
Anti-CD8-PE |
IgG1 |
9012-0087 |
eBioscience, Hatfield, UK |
|
Anti-CD14-PE |
IgG1 |
12-0149-42 |
eBioscience, Hatfield, UK |
|
Anti-CD19-PE |
IgG1 |
12-0198-42 |
eBioscience, Hatfield, UK |
|
Anti-CD45RA-PerCP/Cy5.5 |
IgG2b |
304121 |
Biolegend, London, UK |
|
Anti-CD45RO-PerCP/Cy5.5 |
IgG2a |
304222 |
Biolegend, London, UK |
|
Anti-HLA-G-APC, clone MEM-G/9 |
IgG1 |
A15708 |
Life Technologies, Paisley, UK |
|
Anti-CD56-PE |
IgG1 |
12-0567-42 |
eBioscience, Hatfield, UK |
|
Anti-CD69-PerCP/Cy5.5 |
IgG1 |
310926 |
Biolegend, London, UK |
|
Anti-HLA-DR-FITC |
IgG2b |
327006 |
Biolegend, London, UK |
|
Anti-KIR2DL4-PE |
IgG1 |
347006 |
Biolegend, London, UK |
|
Anti-CD103-FITC |
IgG1 |
350204 |
Biolegend, London, UK |
|
Anti-CD257 (BAFF)-FITC |
IgG1 |
11-9017-42 |
eBioscience, Hatfield, UK |
|
Anti-CD268 (BAFFR)- PerCP/Cy5.5 |
IgG1 |
316918 |
Biolegend, London, UK |
|
Isotype control-PE |
IgG2b |
ab91532 |
AbCam, Cambridge, UK |
|
Isotype control IgG1κ |
IgG1 |
400112 |
Biolegend, London, UK |
|
Isotype control-APC |
IgG1 |
MG105 |
Life Technologies, Paisley, UK |
2.5 Immunosuppressive Drug Treatment
Mononuclear cells were prepared from healthy subjects and resuspended in CM at a concentration of 2 x 106 cells/ml. They were then aliquoted into a 6 well plate and cultured for 7 days at 37ºC, 5% CO2 either with or without 1µg/ml CMV antigen extract (BioRad, Watford, UK), together with one or both of the standard maintenance drugs used for most kidney transplant recipients; cells incubated in CM alone served as controls. Both Prograf® (tacrolimus; Astellas Pharma, Chertsey, UK) and Mycophenolate mofetil (CellSept, Roche, Welwyn Garden City, UK) were obtained in soluble formulation and used at the physiological therapeutic concentrations: Prograf®, 10ng/ml [37] and Mycophenolate mofetil, 3µg/ml [38]. After 7 days’ culture, aliquots of 4 x 105 cells were labelled with a combination of mAb as above and analysed for expression of HLA-G using an Accuri C6 flow cytometer.
2.6 Soluble HLA-G ELISA
Serum samples from healthy subjects and renal transplant patients were stored at -20ºC and assayed together for soluble HLA-G (sHLA-G) using an ELISA kit (Oxford Biosystems, UK) according to the manufacturer’s instructions. Samples were taken from transplant patients immediately prior to transplantation and at intervals of 2, 6 and 12 months post-transplantation, diluted 1:8 with PBS and assayed for sHLA-G.
2.7 Statistical Analysis
Differences in proportions of leucocyte subpopulations expressing HLA-G before and after transplantation were tested using a Wilcoxon matched-pairs signed rank test (two tails). Levels of sHLA-G before and after transplantation were compared using a Mann-Whitney U-test. In all cases, p<0.05 was considered as statistically significant.
3. Results
3.1 Changes in Leucocyte HLA-G Expression Post-Transplantation
HLA-G expression by a range of leucocyte subsets was examined immediately prior to transplantation and at 2 months post-transplant in a group of 21 kidney transplant patients. Proportions of HLA-G+ total CD4+ T cells and CD4+45RA and CD4+45RO subsets pre-transplantation were <0.5% except for CD4+69+ cells (3.6+/-1.2%). A significantly higher proportion of CD4+ T cells expressed HLA-G at 2 months post-transplant and this was also found both for CD4+45RO+ and CD4+45RA+ cells as well as for CD4+69+ subsets (Figure 1).
When a similar comparison was made of CD8+ T cells and their subsets pre-transplantation, <1% of cells were HLA-G+ in total CD8+ and CD8+45RA+ populations, with 2.4+/-0.8% of CD8+45RO+ and 11.1+/1.7% of CD8+69+ cells being positive. All CD8+ T cell subsets showed an increase in proportions of HLA-G+ cells but, unlike for CD4+ T cells, in no cases did this reach statistical significance (Figure 2).
Expression of HLA-G by other leucocyte subsets pre- and 2 months post-transplantation is shown in Figure 3. Proportions of HLA-G+CD56+ T cells were initially high (14.7+/-3.7%) and increased further post-transplantation. Both NK cells (CD3-CD56+) and B cells (CD19+) initially had around 1% of HLA-G+ cells which were increased at 2 months post-transplantation. Around 4% of monocytes were initially HLA-G+, increasing slightly post-transplantation, but in no case did these increases reach statistical significance (Figure 3). Representative flow cytometry plots for CD8+ and CD56+ T cells are shown in Figure 4.
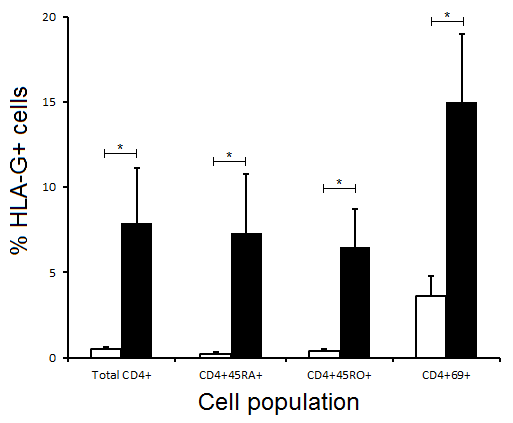
Figure 1 Expression of HLA-G by total CD4+ T cells and subsets thereof from 21 renal transplant patients immediately before (open columns) and 2 months post-transplant (filled columns). Results are expressed as mean +/- SEM; * p<0.01.
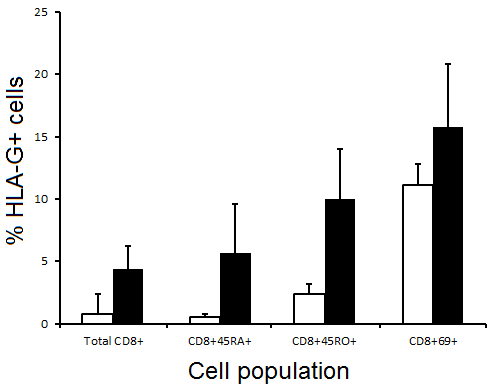
Figure 2 Expression of HLA-G by total CD8+ T cells and subsets thereof from 21 renal transplant patients immediately before (open columns) and 2 months post-transplant (filled columns). Results are expressed as mean +/- SEM; p>0.05 for all groups.
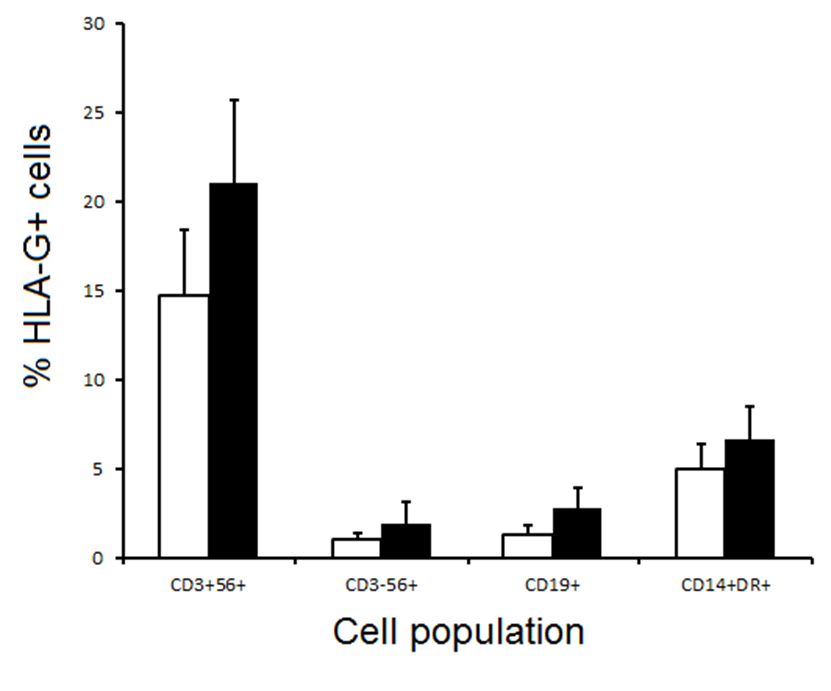
Figure 3 Expression of HLA-G by CD56+ T cells, NK cells, B cells and CD14+HLA-DR+ monocytes from 21 renal transplant patients immediately before (open columns) and 2 months post-transplant (filled columns). Results are expressed as mean +/- SEM; p>0.05 for all groups.
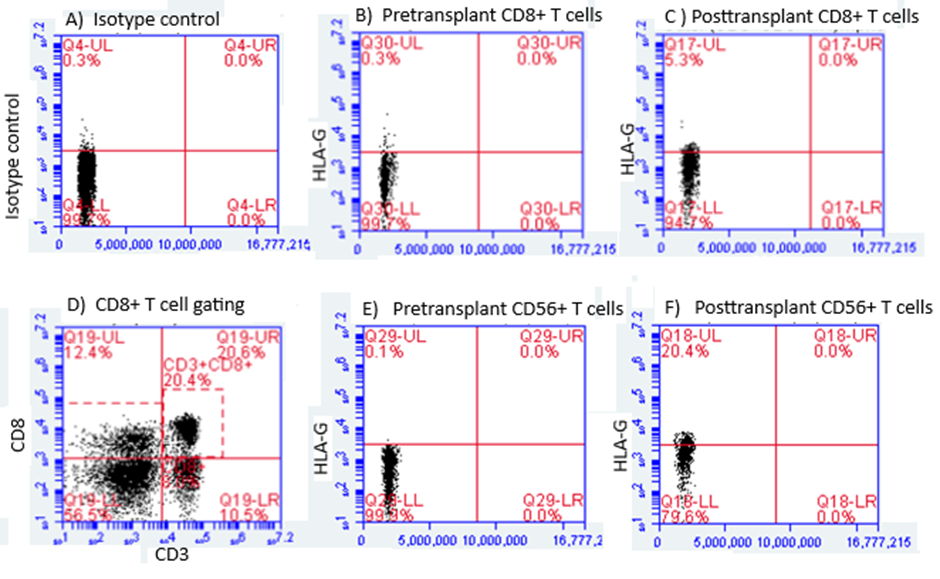
Figure 4 Representative flow cytometry plots for HLA-G expression by CD8+ and CD56+ lymphocytes measured in the same patient pre- and 6 months post-transplantation. A) Isotype control; B) BD8+ T cells pre-transplant; C) CD8+ T cells post-transplant; D) gating strategy for CD8+ T cells; E) CD56+ T cells pre-transplant; F) CD56+ T cells post-transplant.
All patients at risk for CMV infection received antiviral prophylaxis and none of them experienced an episode of CMV viremia, nor did any experience an episode of acute rejection, during the period of the study.
3.2 Changes in Levels of Serum Soluble HLA-G Post-Transplantation
Serum samples were taken from a panel of 30 renal transplant patients immediately prior to and at 6 months post-transplantation. Levels of soluble HLA-G were measured by ELISA, showing a wide range of titers and in a pattern that was not normally distributed. Median levels were increased from 143U/ml (range 59-1255U/ml; n =20) to 246U/ml (range 49-969U/ml; n = 20) at 6 months but, when analysed using a non-parametric method, this increase did not reach statistical significance (Figure 5).

Figure 5 Serum soluble HLA-G concentrations in transplant patients before and at 6 months after transplantation. Mean +/- SEM are shown; p>0.05.
3.3 Effect of Immunosuppressive Drugs on HLA-G Expression
In order to investigate the contribution of the standard maintenance immunosuppressive drug treatment to the observed changes, peripheral blood leucocytes from a panel of 5 healthy subjects were cultured in vitro with Prograf® and Mycophenolate mofetil at the effective concentrations maintained in vivo in transplant patients, in the presence or absence of CMV antigens. Cells cultured in CM for 7 days served as untreated controls. As there was a relatively high incidence of cell death in both treated and untreated populations, viable cells were gated for flow cytometric analysis based upon forward and side scatted properties. With culture in the immunosuppressive drugs alone, total CD4+ and CD8+ T cells showed no change in expression of HLA-G but both CD56+ T cells and B cells showed significantly increased proportions of HLA-G+ cells after 7 days (Figure 6).
NK cells and monocytes both had increased proportions of HLA-G+ cells but in neither case was this statistically significant. Stimulation with CMV antigens in the presence of immunosuppressive drugs resulted in a small but not statistically significant increase in the proportion of HLA-G+ CD4+ T cells compared to stimulation with CMV antigens alone. However, for CD8+ T cells immunosuppressive drugs led to lower proportions of HLA-G+ cells compared to stimulation with CMV antigens alone but in neither case were these changes statistically significant (data not shown).
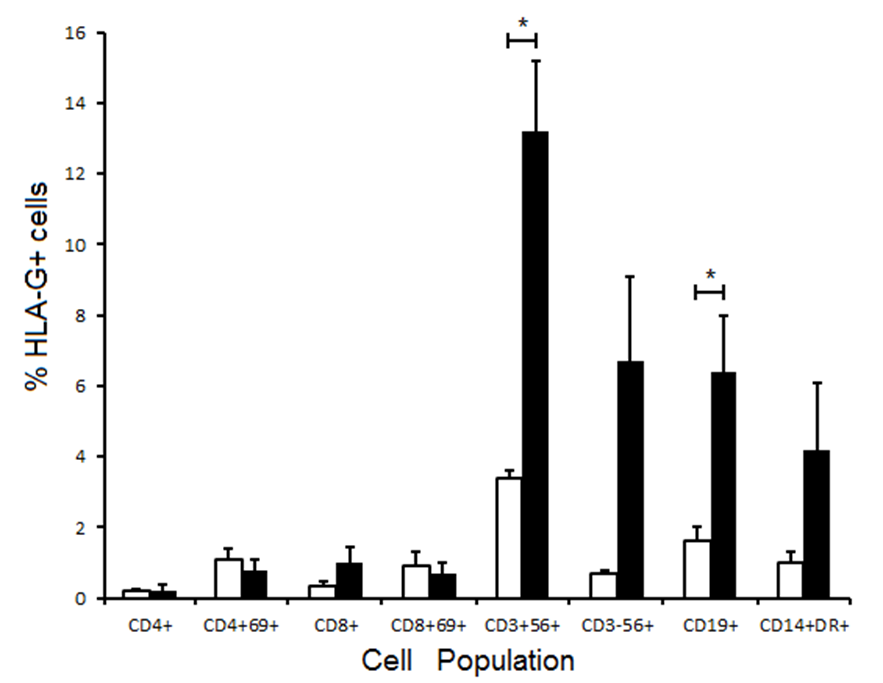
Figure 6 Expression of HLA-G by leucocyte subsets from healthy subjects (n=5) cultured for 7 days with (filled columns) or without Prograf® and mycophenolate mofetil at physiological concentrations (open columns). Results are expressed as mean +/- SEM; * p<0.05.
3.4 Co-expression of HLA-G and CD103 after immunosuppressive drug treatment
The same cell populations were also labelled with anti-CD103 to test for co-expression with HLA-G. Compared to untreated control cells, immunosuppressive drug treatment increased proportions of HLA-G+ cells for all CD103+ cell subsets tested. This was not statistically significant for classical T cell subsets but CD56+103+ T cells, CD103+ NK cells and CD103+ B cells all showed significantly higher proportions of HLA-G+ cells (Figure 7).
Staining of replicate cell samples was performed using anti-CD257 (BAFF) and anti-CD268 (BAFFR) to test for any effect of immunosuppression upon expression of B cell activating factor and its receptor. While there were increases in proportions of all lymphocyte subsets tested expressing both molecules, and also in proportions of HLA-G+ BAFF+ and HLA-G+ BAFFR+ cells, none of these changes were statistically significant (data not shown).
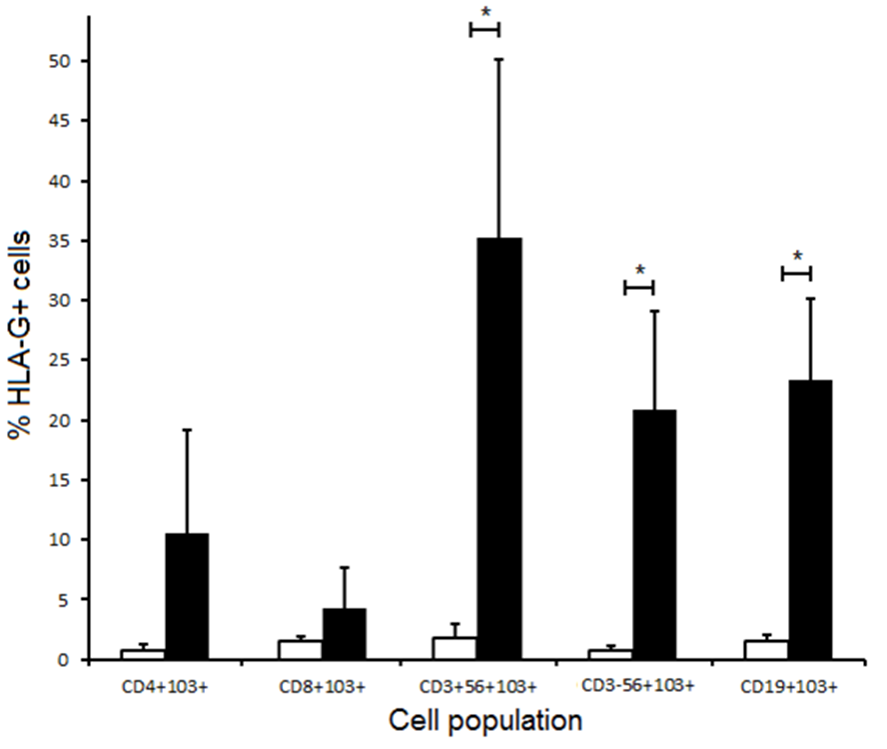
Figure 7 Expression of HLA-G on CD103+ leucocyte subsets from healthy subjects (n=5) cultured for 7 days with (filled columns) or without Prograf® and mycophenolate mofetil at physiological concentrations (open columns). Results are expressed as mean +/-SEM; * p<0.05.
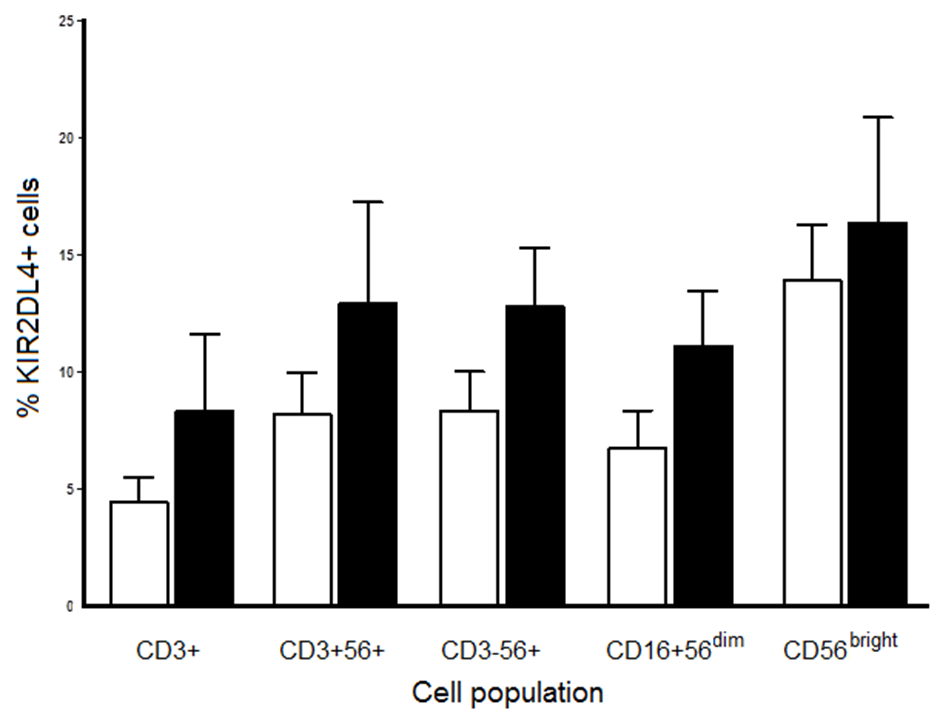
Figure 8 Cell surface expression of KIR2DL4 on NK cell and T cell subsets from renal transplants (n=10) before (open columns) and at 2 months post-transplant (filled columns). Results are expressed as mean +/- SEM; p>0.05.
3.5 Expression of KIR2DL4 in transplant patients
KIR2DL4 is one of the ligands for HLA-G and is expressed mainly on NK cells. A series of 10 patients were tested for its cell surface expression on several NK and T cell subsets immediately prior to and 2 months post-transplantation. In pre-transplant samples, 4.2 +/-1.1% of T cells expressed KIR2DL4, with 7.7 +/-2.3% of CD3+56+ cells expressing it (Figure 8).
Similar proportions of CD56+ NK cells were positive (8.4 +/-3.4%) but a higher proportion of the minor CD56bright subset (14.0 +/-2.2%) was positive (Fig. 6). Matched patients were analysed 2 months following transplantation and in all cell populations tested the proportions of cell surface KIR2DL4+ cells had increased, although none of these changes were statistically significant (p>0.05; Figure 8).
4. Discussion
In the context of clinical transplantation, expression of cell-associated or soluble HLA-G may be regarded as advantageous in prevention of acute rejection of transplanted organs. The significantly increased proportions of HLA-G+ CD4+ T cell subsets, including naïve, memory and activated cells, post-transplantation would potentially be able to mediate immune suppression of cells expressing ligands for HLA-G. This could be a result of selective proliferation of the small numbers of HLA-G+ cells or induction of HLA-G expression by previously negative cells. A proportion of the HLA-G+ CD4+ T cells, particularly of the CD69+ subset, may have been Tregs and it has been suggested that sHLA-G may participate in graft acceptance through induction of Tregs [39]. In our previous work with healthy subjects, only around 1-2% of Tregs expressed HLA-G and this proportion was decreased further following stimulation with CMV antigens [36]. Alternative possibilities are that HLA-G CD4 T cells are a component of the alloreactive population or are induced following immunosuppressive drug treatment. Previous work has shown a significant increase in proportions of HLA-G+ CD4+ T cells post-renal transplantation in patients with stable graft function [39]. Furthermore, these cells were able to suppress a mixed lymphocyte reaction, indicative of a contribution to graft survival [40]. HLA-G expressed by dendritic cells or in soluble form can downregulate CD4 and CD8 on allostimulated T cells, leading to generation of suppressor function [41]. Both cell-associated [42] and soluble HLA-G are capable of suppressing alloresponses [43]. Alternatively, increases in HLA-G expression post-transplantation could be the result of re-activation of latent viruses, such as CMV or BK virus, although none of the patients studied showed any detectable viremia during the observation period.
However, when cells from healthy subjects were incubated for 7 days in the presence of the standard immunosuppressive drugs used in kidney transplant patients, at therapeutic doses, no change in proportions of HLA-G+ CD4+ T cells was noted. This would be supportive of the possibility that allostimulation of CD4+ T cells leads to induction of HLA-G expression or expansion of a minor HLA-G+ CD4+ T cell population. There were, however, significant increases in proportions of HLA-G+ CD56+ T cells and B cells with immunosuppressive drug treatment and these could potentially contribute to production of soluble HLA-G. However, HLA-G detectable at the cell surface by monoclonal antibodies may not have been produced by those cells but could potentially have been taken up from other cells; mRNA analysis would be needed to ascertain the cellular source.
Proportions of both CD56+ T cells and B cells expressing HLA-G were significantly increased following stimulation in vitro of leucocytes from healthy subjects with CMV antigens [36] and also in kidney transplant patients in vivo [44]. It remains possible that immunosuppressive drug treatment of leucocytes in vitro permitted sub-clinical emergence of latent CMV in positive subjects, leading to enhancement of HLA-G expression. All at-risk renal transplant patients were treated with anti-CMV prophylaxis, which minimised the possibility of such CMV reactivation occurring in vivo during the first two months post-transplantation and was not noted in any of the study subjects.
Although median levels of sHLA-G were not significantly increased in the present experiments, previous reports have described increases post-transplantation that were associated with graft acceptance [8,45,46,47,48]. Serum rather than plasma samples were exclusively used in the present experiments and, as it has been reported that sHLA-G can absorb to fibrin clots [49], it remains possible that higher levels might have been found in plasma samples. Alloreactive CD4+ T cells have been reported to secrete sHLA-G, thereby suppressing the alloreactive response [50]. However, none of the patients in the present study experienced an episode of acute rejection within the first 2 months post-transplantation. Everolimus has been reported to upregulate HLA-G expression in heart transplant patients [51], specifically by enhancing synthesis by human coronary artery smooth muscle cells, and may be implicated in heart transplant acceptance [52].
The mucosal leucocyte integrin CD103 (αEβ7) has previously been reported to be expressed on CD8+ T cells mediating allograft rejection [53] but also on CD8+ regulatory T cells [54]. When co-expression of HLA-G and CD103 was examined in CD56+ T cells, NK cells and B cells from healthy subjects cultured with immunosuppressive drugs, significant increases were found compared to untreated controls. This could be a result of either selective expansion of the small subsets of HLA-G+CD103+ cells or induction of these markers on previously negative cells. In previous studies, rapamycin was found to enhance the numbers of CD103+CD8+ regulatory T cells [55] whereas tacrolimus reduced CD103+ expression in chronic graft-versus-host responses in vivo [56]. B cells do not normally express CD103 [57,58] so the increased expression seen with immunosuppressive drugs in vitro may alter the migration pattern of some B cells if it occurs in vivo. Expression of HLA-G on CD103+ cells in vivo could potentially lead to inhibition of ILT2, ILT4 or KIR2DL4 positive cells.
Any immunosuppressive role of cell-associated or soluble HLA-G in clinical transplantation would be dependent upon expression of ligands for HLA-G. When expression of KIR2DL4 was examined prior to and at 2 months post transplantation, higher proportions of all lymphocyte subsets tested were noted, although in no case did this reach statistical significance. Cyclosporin A has been reported to induce ILT4 expression by a human NK cell line [59] and rapamycin induced dendritic cells expressing high levels of ILT4 [60], although in another report it was found to have the opposite effect on dendritic cell ILT2 and 4 expression [61]. However, in the latter case this led to T cell hyporesponsiveness [60]. Functionally, binding of HLA-G to ILT2-expressing B cells led to their inhibition [62].
While upregulation of HLA-G expression in viral infection or malignancy would be disadvantageous to the host, in clinical transplantation it is potentially beneficial for long-term graft acceptance. It would be of interest to characterise more fully any changes in expression of HLA-G or its ligands following alloreactive responses to determine the feasibility of suppressing alloreactivity therapeutically.
Abbreviations
CM, complete medium; CMV, cytomegalovirus; HLA, human leucocyte antigen/s; ILT, immunoglobulin-like transcript; KIR, killer immunoglobulin-like receptor; LILR, leucocyte immunoglobulin-like receptor; mAb, monoclonal antibody; NK, natural killer; PBS, phosphate buffered saline; sHLA-G, soluble HLA-G.
Acknowledgments
ZA was in receipt of a studentship from the Iraqi Cultural Bureau.
Author Contributions
AH and SEC conceived the study; ZA conducted the experimental work and analysed the data; SH and JM coordinated patient sample collection; BF supervised genomic analysis; DM advised on KIR investigations. SEC wrote the manuscript with input from all other co-authors.
Competing Interests
The authors have declared that no competing interests exist.
References
- Hammer A, Hutter H, Dohr G. HLA class I expression on the materno-fetal interface. Am J Reprod Immunol. 1997; 38: 150-157. [CrossRef]
- Ferreira LM, Meissner TB, Tilburgs T, Strominger JL. HLA-G: At the interface of maternal-fetal tolerance. Trends Immunol. 2017; 38: 272-286. [CrossRef]
- LeMaoult J, Caumartin J, Daouya M, Favier B, Le Rond S, Gonzalez A, et al. Immune regulation by pretenders: cell-to-cell transfers of HLA-G make effector T cells act as regulatory cells. Blood. 2007: 109: 2040-2048. [CrossRef]
- Carosella ED, Rouas-Freiss N, Tronik-Le Roux D, Moreau P, LeMaoult J. HLA-G: An immune checkpoint molecule. Adv Immunol. 2015; 127: 33-144. [CrossRef]
- Clements CS, Kjer-Nielsen L, McCluskey J, Rossjohn J. Structural studies on HLA-G: implications for ligand and receptor binding. Hum Immunol. 2007; 68: 220-226. [CrossRef]
- Lila N, Carpentier A, Amrein C, Khalil-Daher I, Dausset J, Carosella ED. Implication of HLA-G molecule in heart-graft acceptance. Lancet. 2000; 355: 2138. [CrossRef]
- Lila N, Amrein C, Guillemain R, Chevalier P, Latremouille C, Fabiani J, et al. Human leukocyte antigen-G expression after heart transplantation is associated with a reduced incidence of rejection. Circulation. 2002; 105: 1949-1954. [CrossRef]
- Luque J, Torres MI, Aumente MD, Lozano JM, García-Jurado G, González R, et al. sHLA-G levels in the monitoring of immunosuppressive therapy and rejection following heart transplantation. Transpl Immunol. 2006; 17: 70-73. [CrossRef]
- Blanco-García RM, López-Álvarez MR, Garrido IP, Salgado-Cecilia G, Campillo JA, Bolarín JM, et al. Post-transplant increase in soluble human leukocyte antigen-G associated with non-severe cardiac allograft vasculopathy. Hum Immunol. 2013; 74: 318-324. [CrossRef]
- Almasood A, Sheshgiri R, Joseph JM, Rao V, Kamali M, Tumiati L, et al. Human leukocyte antigen-G is upregulated in heart failure patients: a potential novel biomarker. Hum Immunol. 2011; 72: 1064-1067. [CrossRef]
- Brugière O, Thabut G, Pretolani M, Krawice‐Radanne I, Dill C, Herbreteau A, et al. Immunohistochemical study of HLA-G expression in lung transplant recipients. Am J Transplant. 2009; 9: 1427-1438. [CrossRef]
- Brugière O, Thabut G, Krawice-Radanne I, Rizzo R, Dauriat G, Danel C, et al. Role of HLA-G as a predictive marker of low risk of chronic rejection in lung transplant recipients: a clinical prospective study. Am J Transplant. 2015; 15: 461-471. [CrossRef]
- Zarkhin V, Talisetti A, Li L, Wozniak LG, McDiarmid SV, Cox K, et al. Expression of soluble HLA-G identifies favorable outcomes in liver transplant recipients. Transplantation. 2010; 90: 1000–1005. [CrossRef]
- Racca AL, Veaute CM, Bailat AS, Gaite L, Arriola M, Hajos SE, et al. Expression of HLA-G and MICA mRNA in renal allograft. Transpl Immunol. 2009; 21: 10-12. [CrossRef]
- Bi LL, Gao Y, Sun YJ, Kong XR, He XY, Ma XH, et al. Research of mHLA-G and the receptor expression with kidney rejection and CMV active infection following kidney transplantation. Zhonghua Yi Xue Za Zhi. 2017; 97: 104-107.
- Žilinská Z, Bandžuchová H, Chrastina M, Trebaticky B, Breza J, Handzušová M, et al. Expression of HLA-G transcripts in graft biopsy samples of renal transplant recipients. Transpl Immunol. 2015; 33: 159-165. [CrossRef]
- Crispim JC, Duarte RA, Soares CP, Costa R, Silva JS, Mendes-Júnior CT, et al. Human leukocyte antigen-G expression after kidney transplantation is associated with a reduced incidence of rejection. Transpl Immunol. 2008; 18: 361-367. [CrossRef]
- Cilião Alves DC, de Oliveira Crispim JC, Castelli EC, Mendes-Junior CT, Deghaide NHS, Silva GEB, et al. Human leukocyte antigen-G 3' untranslated region polymorphisms are associated with better kidney allograft acceptance. Hum Immunol. 2012; 73: 52-59. [CrossRef]
- Jin ZK, Xu CX, Tian PX, Xue WJ, Ding XM, Zheng J, et al. Impact of HLA-G 14-bp polymorphism on acute rejection and cytomegalovirus infection in kidney transplant recipients from northwestern China. Transpl Immunol. 2012; 27: 69-74. [CrossRef]
- Misra MK, Prakash S, Kapoor R, Pandey SK, Sharma RK, Agrawal S. Association of HLA-G promoter and 14-bp insertion-deletion variants with acute allograft rejection and end-stage renal disease. Tissue Antigens. 2013; 82: 317-326. [CrossRef]
- Crispim JC, Mendes-Junior CT, Wastowski IJ, Costa R, Castelli EC, Saber LT, et al. Frequency of insertion/deletion polymorphism in exon 8 of HLA-G and kidney allograft outcome. Tissue Antigens. 2008; 71: 35-41.
- Hou W, Huang Z, Ji Z, Zhou J. Human leukocyte antigen-G-14-base-pair-insertion/deletion polymorphism and graft survival in kidney transplant recipients. Exp Clin Transplant. 2014; 12: 89-94.
- Littera R, Piredda G, Pani A, Frongia M, Onano B, Michittu MB, et al. Role of human leukocyte antigen-G 14-base pair polymorphism in kidney transplantation outcomes. J Nephrol. 2013; 26: 1170-1178. [CrossRef]
- Thude H, Janssen M, Sterneck M, Nashan B, Koch M. 14-bp ins/del polymorphism and +3142C>G SNP of the HLA-G gene have a significant impact on acute rejection after liver transplantation. Hum Immunol. 2016; 77: 1159-1165. [CrossRef]
- Pirri A, Contieri FC, Benvenutti R, Bicalho-Mda G. A study of HLA-G polymorphism and linkage disequilibrium in renal transplant patients and their donors. Transpl Immunol. 2009; 20: 143-149. [CrossRef]
- Qiu J, Terasaki PI, Miller J, Mizutani K, Cai J, Carosella ED. Soluble HLA-G expression and renal graft acceptance. Am J Transplant. 2006; 6: 2152-2156. [CrossRef]
- Kaneku H. Detection of soluble HLA-G and its correlation with kidney transplant outcome. Clin Transpl. 2006; 38; 447-454.
- Rebmann V, Bartsch D, Wunsch A, Möllenbeck P, Golda T, Viebahn R, et al. Soluble total human leukocyte antigen class I and human leukocyte antigen-G molecules in kidney and kidney/pancreas transplantation. Hum Immunol. 2009; 70: 995-999. [CrossRef]
- Jin HL, Li CR, Xiao L, Shi BY, Cai M, Li ZL, et al. Clinical relevance of sHLA-G-mediated with better graft acceptance in early posttransplantation. Transplant Proc. 2012; 44: 1259-1261. [CrossRef]
- Misra MK, Pandey SK, Kapoor R, Sharma RK, Kapoor R, Prakash S, et al. HLA-G gene expression influenced at allelic level in association with end stage renal disease and acute allograft rejection. Hum Immunol. 2014; 75: 833-889. [CrossRef]
- Farid E, Al-Wedaie F, Tabbara K, El-Agroudy AE, Al-Ghareeb SM. Human leukocyte antigen G and renal allograft transplant. Exp Clin Transplant. 2015; 13: 371-376.
- Poláková K, Bandžuchová H, Žilinská Z, Chreňová S, Kuba D, Russ G. Analysis of HLA-G expression in serum and biopsy samples of kidney transplant recipients. Immunobiology. 2015; 220: 533-537. [CrossRef]
- Ezeakile M, Portik-Dobos V, Wu J, Horuzsko DD, Kapoor R, Jagadeesan M, et al. HLA-G dimers in the prolongation of kidney allograft survival. J Immunol Res. 2014; 2014: 153981. [CrossRef]
- Krongvorakul J, Kantachuvesiri S, Ingsathit A, Rattanasiri S, Mongkolsuk T, Kitpoka P, et al. Association of soluble human leukocyte antigen-G with acute tubular necrosis in kidney transplant recipients. Asian Pac J Allergy Immunol. 2015; 33: 117-122.
- S. Le Rond, J. Le Maoult, C. Créput, Menier C, Deschamps M, Le Friec G, et al. Alloreactive CD4+ and CD8+ T cells express the immunotolerant HLA-G molecule in mixed lymphocyte reactions: in vivo implications in transplanted patients. Eur J Immunol. 2004; 34: 649-660. [CrossRef]
- Albayati Z, Alyami A, Alomar S, Middleton D, Bonnett L, Aleem S, et al. The Influence of cytomegalovirus on expression of HLA-G and its ligand KIR2DL4 by human peripheral blood leucocyte subsets. Scand J Immunol. 2017; 86: 396-407. [CrossRef]
- Andrews LM, Li Y, De Winter BCM, Shi YY, Baan CC, Van Gelder T, et al. Pharmacokinetic considerations related to therapeutic drug monitoring of tacrolimus in kidney transplant patients. Expert Opin Drug Metab Toxicol. 2017; 13: 1225-1236. [CrossRef]
- Staatz CE, Tett SE. Clinical pharmacokinetics and pharmacodynamics of mycophenolate in solid organ transplant recipients. Clin Pharmacokinet. 2007; 46: 13-58. [CrossRef]
- S. Le Rond, C. Azéma, I. Krawice-Radanne, Durrbach A, Guettier C, Carosella ED, et al. Evidence to support the role of HLA-G5 in allograft acceptance through induction of immunosuppressive/regulatory T cells. J Immunol. 2006; 176: 3266-3276. [CrossRef]
- Lu N, Zhang Y, Zou X, Yang X, Tian J, Zhen J, et al. HLA-G on peripheral blood CD4(+) T lymphocytes: a potential predictor for acute renal rejection. Transpl Int. 2011; 24: 1103-1111. [CrossRef]
- Naji A, Le Rond S, Durrbach A, Krawice-Radanne I, Creput C, Daouya M, et al. CD3+CD4low and CD3+CD8low are induced by HLA-G: novel human peripheral blood suppressor T-cell subsets involved in transplant acceptance. Blood. 2007; 110: 3936-3948. [CrossRef]
- Riteau B, Menier C, Khalil-Daher I, Sedlik C, Dausset J, Rouas-Freiss N, et al. HLA-G inhibits the allogeneic proliferative response. J Reprod Immunol. 1999; 43: 203-211. [CrossRef]
- Bahri R, Hirsch F, Josse A, Rouas-Freiss N, Bidere N, Vasquez A, et al. Soluble HLA-G inhibits cell cycle progression in human alloreactive T lymphocytes. J Immunol. 2006; 176: 1331-1339. [CrossRef]
- Almehmadi M, Hammad A, Heyworth S, Moberly J, Middleton D, Hopkins MJ, et al. CD56+ T cells are increased in kidney transplant patients following cytomegalovirus infection. Transpl Infect Dis. 2015; 17: 518-526. [CrossRef]
- Creput C, Le Friec G, Bahri R, Amiot L, Charpentier B, Carosella E, et al. Detection of HLA-G in serum and graft biopsy associated with fewer acute rejections following combined liver-kidney transplantation: possible implications for monitoring patients. Hum Immunol. 2003; 64: 1033-1038. [CrossRef]
- Bahri R, Naji A, Menier C, Charpentier B, Carosella ED, Rouas-Freiss N, et al. Dendritic cells secrete the immunosuppressive HLA-G molecule upon CTLA4-Ig treatment: implications in human renal transplant acceptance. J Immunol. 2009; 183: 7054-7062. [CrossRef]
- Jin Z, Xu C, Duan W, Yang J, Tian P. Serum soluble HLA-G, soluble CD30 is correlated to the time after transplantation in renal transplant recipients. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2017; 33: 947-952.
- Pabón MA, Navarro CE, Osorio JC, Gómez N, Moreno JP, Donado AF, et al. Impact of human leukocyte antigen molecules E, F and G on the outcome of transplantation. Transplant Proc. 2014; 46: 2957-2965. [CrossRef]
- Rudstein-Svetlicky N, Loewenthal R, Horejsi V, Gazit E. HLA-G levels in serum and plasma. Tissue Antigens. 2006; 67: 111-116. [CrossRef]
- Lila N, Rouas-Freiss N, Dausset J, Carpentier A, Carosella ED. Soluble HLA-G protein secreted by allo-specific CD4+ T cells suppresses the allo-proliferative response: a CD4+ T cell regulatory mechanism. Proc Natl Acad Sci USA. 2001; 98: 12150-12155. [CrossRef]
- Sheshgiri R, Gustafsson F, Sheedy J, Rao V, Ross HJ, Delgado DH. Everolimus but not mycophenolate mofetil therapy is associated with soluble HLA-G expression in heart transplant patients. J Heart Lung Transplant. 2009; 28: 1193-1197. [CrossRef]
- Mociornita AG, Tumiati L, Joseph JL, et al., Everolimus induces human leukocyte antigen-G expression in human coronary artery smooth muscle cells: Impact on graft rejection and cardiac allograft vasculopathy. Circulation. 2011; 124: A13205.
- Yuan R, El-Asady R, Liu K, Wang D, Drachenberg CB, Hadley GA. Critical role for CD103+CD8+ effectors in promoting tubular injury following allogeneic renal transplantation. J Immunol. 2005; 175: 2868-2879. [CrossRef]
- Koch SD, Uss E, van Lier RA, ten Berge IJM. Alloantigen-induced regulatory CD8+CD103+ T cells. Hum Immunol. 2008; 69: 737-744. [CrossRef]
- Uss E, Yong SL, Hooibrink B, van Lier RAW, ten Berge IJM. Rapamycin enhances the number of alloantigen-induced human CD103+CD8+ regulatory T cells in vitro. Transplantation. 2007; 83: 1098-1106. [CrossRef]
- Motta A, Zhan Q, Larson A, Lerman M, Woo SB, Soiffer RJ, et al. Immunohistopathological characterization and the impact of topical immunomodulatory therapy in oral chronic graft-versus-host disease: A pilot study. Oral Dis. 2018; 24: 580-590. [CrossRef]
- Höffkes HG, Schmidtke G, Uppenkamp M, Schmücker U. Multiparametric immunophenotyping of B cells in peripheral blood of healthy adults by flow cytometry. Clin Diagn Lab Immunol. 1996; 3: 30-36.
- Deneys V, Mazzon AM, Marques JM, Benoit H, De Bruyère M. Reference values for peripheral blood B-lymphocyte subpopulations: a basis for multiparametric immunophenotyping of abnormal lymphocytes. J Immunol Methods. 2001; 253: 23-36. [CrossRef]
- Si YQ, Bian XK, Lu N, Jia YF, Hou ZH, Zhang Y. Cyclosporine induces up-regulation of immunoglobulin-like transcripts 3 and 4 expression on and activity of NKL cells. Transplant Proc. 2012; 44: 1407-1411. [CrossRef]
- Stallone G, Pontrelli P, Infante B, Gigante M, Netti GS, Ranieri E, et al. Rapamycin induces ILT3(high)ILT4(high) dendritic cells promoting a new immunoregulatory pathway. Kidney Int. 2014; 85: 888-897. [CrossRef]
- Fedoric B, Krishnan R. Rapamycin downregulates the inhibitory receptors ILT2, ILT3, ILT4 on human dendritic cells and yet induces T cell hyporesponsiveness independent of FoxP3 induction. Immunol Lett. 2008; 120: 49-56. [CrossRef]
- Naji A, Menier C, Morandi F, Agaugué S, Maki G, Ferretti E, et al. Binding of HLA-G to ITIM-bearing Ig-like transcript 2 receptor suppresses B cell responses. J Immunol. 2014; 192: 1536-1546. [CrossRef]


